CP-31398 restores mutant p53 tumor suppressor function and inhibits UVB-induced skin carcinogenesis in mice
- PMID: 18060030
- PMCID: PMC2096455
- DOI: 10.1172/JCI32481
CP-31398 restores mutant p53 tumor suppressor function and inhibits UVB-induced skin carcinogenesis in mice
Abstract
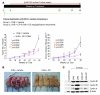
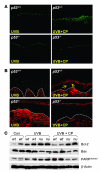
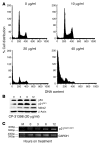
Mutations in the tumor suppressor p53 are detectable in over 50% of all human malignancies. Mutant p53 protein is incapable of transactivating its downstream target genes that are required for DNA repair and apoptosis. Chronic exposure to UVB induces p53 mutations and is carcinogenic in both murine and human skin. CP-31398, a styrylquinazoline compound, restores the tumor suppressor functions of mutant forms of p53 in tumor cells. However, its effectiveness in vivo remains unclear. Here, we demonstrate that CP-31398 blocked UVB-induced skin carcinogenesis and was associated with increases in p53, p21, and BclXs. CP-31398 downregulated Bcl2, proliferating nuclear cell antigen, and cyclin D1. Activation of caspase-3 and cleavage of poly (ADP-ribose) polymerase also occurred in both tumor and perilesional skin following treatment. CP-31398 induced the expression of p53-dependent target proteins, and this was followed by apoptosis in UVB-irradiated wild-type mice but not in their p53-deficient littermates. Similar effects were observed in human skin carcinoma A431 cells expressing mutant p53. In addition, CP-31398 induced mitochondrial translocation of p53, leading to changes in mitochondrial membrane permeability pore transition (MPT) and consequent cytochrome c release in these cells. Blocking MPT diminished p53 translocation and apoptosis. These studies indicate that reconstituting p53 tumor suppressor functions in vivo by small molecular weight compounds may block the pathogenesis and progression of skin cancer.
Figures
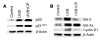

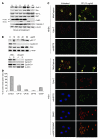
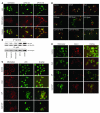
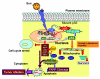
Comment in
-
Targeting mutant p53 shows promise for sunscreens and skin cancer.J Clin Invest. 2007 Dec;117(12):3658-60. doi: 10.1172/JCI34251. J Clin Invest. 2007. PMID: 18060027 Free PMC article.
References
-
- Vogelstein B., Kinzler K.W. Cancer genes and the pathways they control. Nat. Med. 2004;10:789–799. - PubMed
-
- Vogelstein B., Lane D., Levine A.J. Surfing the p53 network. Nature. 2000;408:307–310. - PubMed
-
- van Kranen H.J., de Gruijl F.R. Mutations in cancer genes of UV-induced skin tumors of hairless mice. J. Epidemiol. 1999;9:S58–S65. - PubMed
-
- Phan T.A., Halliday G.M., Barnetson R.S., Damian D.L. Spectral and dose dependence of ultraviolet radiation-induced immunosuppression. Front. Biosci. 2006;11:394–411. - PubMed
-
- D’Errico M., Lemma T., Calcagnile A., Santis L.P.D., Dogliotti E. Cell type and DNA damage specific response of human skin cells to environmental agents. Mutat. Res. 2007;614:37–47. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

