Adoptive transfer of effector CD8+ T cells derived from central memory cells establishes persistent T cell memory in primates
- PMID: 18060041
- PMCID: PMC2104476
- DOI: 10.1172/JCI32103
Adoptive transfer of effector CD8+ T cells derived from central memory cells establishes persistent T cell memory in primates
Abstract
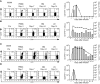
The adoptive transfer of antigen-specific T cells that have been expanded ex vivo is being actively pursued to treat infections and malignancy in humans. The T cell populations that are available for adoptive immunotherapy include both effector memory and central memory cells, and these differ in phenotype, function, and homing. The efficacy of adoptive immunotherapy requires that transferred T cells persist in vivo, but identifying T cells that can reproducibly survive in vivo after they have been numerically expanded by in vitro culture has proven difficult. Here we show that in macaques, antigen-specific CD8(+) T cell clones derived from central memory T cells, but not effector memory T cells, persisted long-term in vivo, reacquired phenotypic and functional properties of memory T cells, and occupied memory T cell niches. These results demonstrate that clonally derived CD8+ T cells isolated from central memory T cells are distinct from those derived from effector memory T cells and retain an intrinsic capacity that enables them to survive after adoptive transfer and revert to the memory cell pool. These results could have significant implications for the selection of T cells to expand or to engineer for adoptive immunotherapy of human infections or malignancy.
Figures



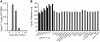


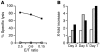
References
-
- Cheever M.A., Greenberg P.D., Fefer A. Specificity of adoptive chemoimmunotherapy of established syngeneic tumors. J. Immunol. 1980;125:711–714. - PubMed
-
- Riddell S.R., et al. Restoration of viral immunity in immunodeficient humans by the adoptive transfer of T cell clones. Science. 1992;257:238–241. - PubMed
-
- Walter E.A., et al. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N. Engl. J. Med. 1995;333:1038–1044. - PubMed
-
- Rooney C.M., et al. Infusion of cytotoxic T cells for the prevention and treatment of Epstein-Barr virus-induced lymphoma in allogeneic transplant recipients. Blood. 1998;92:1549–1555. - PubMed
Publication types
MeSH terms
Grants and funding
- M01RR00037/RR/NCRR NIH HHS/United States
- P30CA33572-21/CA/NCI NIH HHS/United States
- R01 AI053193/AI/NIAID NIH HHS/United States
- R01AI053193/AI/NIAID NIH HHS/United States
- R01 CA114536/CA/NCI NIH HHS/United States
- CA107399/CA/NCI NIH HHS/United States
- P30 CA033572/CA/NCI NIH HHS/United States
- CA30206-25A1/CA/NCI NIH HHS/United States
- P01 CA030206/CA/NCI NIH HHS/United States
- P50 CA107399/CA/NCI NIH HHS/United States
- R01CA114536/CA/NCI NIH HHS/United States
- CA18029/CA/NCI NIH HHS/United States
- U01HL66947/HL/NHLBI NIH HHS/United States
- P01 CA018029/CA/NCI NIH HHS/United States
- M01 RR000037/RR/NCRR NIH HHS/United States
- U01 HL066947/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

