The calcium channel alpha2/delta1 subunit is involved in extracellular signalling
- PMID: 18063658
- PMCID: PMC2375616
- DOI: 10.1113/jphysiol.2007.147959
The calcium channel alpha2/delta1 subunit is involved in extracellular signalling
Abstract
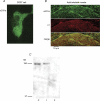
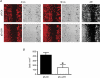
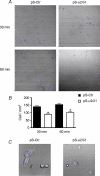
The alpha2/delta1 subunit forms part of the dihydropyridine receptor, an essential protein complex for excitation-contraction (EC) coupling in skeletal muscle. Because of the lack of a viable knock-out animal, little is known regarding the role of the alpha2/delta1 subunit in EC coupling or in other cell functions. Interestingly, the alpha2/delta1 appears before the alpha1 subunit in development and contains extracellular conserved domains known to be important in cell signalling and inter-protein interactions. These facts raise the possibility that the alpha2/delta1 subunit performs vital functions not associated with EC coupling. Here, we tested the hypothesis that the alpha2/delta1 subunit is important for interactions of muscle cells with their environment. Using confocal microscopy, we followed the immunolocalization of alpha2/delta1 and alpha1 subunits with age. We found that in 2-day-old myotubes, the alpha2/delta1 subunit concentrated towards the ends of the cells, while the alpha1 subunit clustered near the centre. As myotubes aged (6-12 days), the alpha2/delta1 became evenly distributed along the myotubes and co-localized with alpha1. When the expression of alpha2/delta1 was blocked with siRNA, migration, attachment and spreading of myoblasts were impaired while the L-type calcium current remained unaffected. The results suggest a previously unidentified role of the alpha2/delta1 subunit in skeletal muscle and support the involvement of this protein in extracellular signalling. This new role of the alpha2/delta1 subunit may be crucial for muscle development, muscle repair and at times in which myoblast attachment and migration are fundamental.
Figures



Comment in
-
Does the voltage-gated calcium channel alpha2delta-1 subunit play a dual function in skeletal muscle?J Physiol. 2008 Apr 15;586(8):2035-7. doi: 10.1113/jphysiol.2008.152298. Epub 2008 Feb 21. J Physiol. 2008. PMID: 18292126 Free PMC article. Review. No abstract available.
References
-
- Alden KJ, Garcia J. Dissociation of charge movement from calcium release and calcium current in skeletal myotubes by gabapentin. Am J Physiol Cell Physiol. 2002;283:C941–C949. - PubMed
-
- Anantharaman V, Aravind L. Cache – a signaling domain common to Ca2+ channel subunits and a class of prokaryotic chemotaxic receptors. Trends Biochem Sci. 2000;25:535–537. - PubMed
-
- Bidaud I, Montiel A, Nargeot J, Lory P. Properties and role of voltage-dependent calcium channels during mouse skeletal muscle differentiation. J Muscle Res Cell Motility. 2006;27:75–81. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

