Beta-catenin defines head versus tail identity during planarian regeneration and homeostasis
- PMID: 18063757
- PMCID: PMC2755502
- DOI: 10.1126/science.1150029
Beta-catenin defines head versus tail identity during planarian regeneration and homeostasis
Abstract
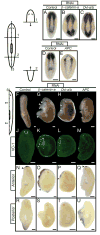
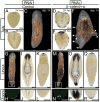
After amputation, freshwater planarians properly regenerate a head or tail from the resulting anterior or posterior wound. The mechanisms that differentiate anterior from posterior and direct the replacement of the appropriate missing body parts are unknown. We found that in the planarian Schmidtea mediterranea, RNA interference (RNAi) of beta-catenin or dishevelled causes the inappropriate regeneration of a head instead of a tail at posterior amputations. Conversely, RNAi of the beta-catenin antagonist adenomatous polyposis coli results in the regeneration of a tail at anterior wounds. In addition, the silencing of beta-catenin is sufficient to transform the tail of uncut adult animals into a head. We suggest that beta-catenin functions as a molecular switch to specify and maintain anteroposterior identity during regeneration and homeostasis in planarians.
Figures


References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

