Secretion of flavins by Shewanella species and their role in extracellular electron transfer
- PMID: 18065612
- PMCID: PMC2227709
- DOI: 10.1128/AEM.01387-07
Secretion of flavins by Shewanella species and their role in extracellular electron transfer
Abstract
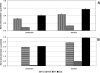
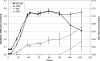
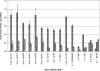
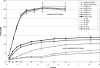
Fe(III)-respiring bacteria such as Shewanella species play an important role in the global cycle of iron, manganese, and trace metals and are useful for many biotechnological applications, including microbial fuel cells and the bioremediation of waters and sediments contaminated with organics, metals, and radionuclides. Several alternative electron transfer pathways have been postulated for the reduction of insoluble extracellular subsurface minerals, such as Fe(III) oxides, by Shewanella species. One such potential mechanism involves the secretion of an electron shuttle. Here we identify for the first time flavin mononucleotide (FMN) and riboflavin as the extracellular electron shuttles produced by a range of Shewanella species. FMN secretion was strongly correlated with growth and exceeded riboflavin secretion, which was not exclusively growth associated but was maximal in the stationary phase of batch cultures. Flavin adenine dinucleotide was the predominant intracellular flavin but was not released by live cells. The flavin yields were similar under both aerobic and anaerobic conditions, with total flavin concentrations of 2.9 and 2.1 micromol per gram of cellular protein, respectively, after 24 h and were similar under dissimilatory Fe(III)-reducing conditions and when fumarate was supplied as the sole electron acceptor. The flavins were shown to act as electron shuttles and to promote anoxic growth coupled to the accelerated reduction of poorly crystalline Fe(III) oxides. The implications of flavin secretion by Shewanella cells living at redox boundaries, where these mineral phases can be significant electron acceptors for growth, are discussed.
Figures

References
-
- Beliaev, A. S., D. A. Saffarini, J. L. McLaughlin, and D. Hunnicutt. 2001. MtrC, an outer membrane decahaem c cytochrome required for metal reduction in Shewanella putrefaciens MR-1. Mol. Microbiol. 39:722-730. - PubMed
-
- Brettar, I., R. Christen, and M. G. Hofle. 2002. Shewanella denitrificans sp. nov., a vigorously denitrifying bacterium isolated from the oxic-anoxic interface of the Gotland Deep in the central Baltic Sea. Int. J. Syst. Evol. Microbiol. 52:2211-2217. - PubMed
-
- Conn, E. E., and P. K. Stumpf. 1976. Outlines of biochemistry, 4th ed. John Wiley & Sons, New York, NY.
-
- Demain, A. L. 1972. Riboflavin oversynthesis. Annu. Rev. Microbiol. 26:369-388. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

