Adjuvant effect of anti-4-1BB mAb administration in adoptive T cell therapy of cancer
- PMID: 18071585
- PMCID: PMC2096735
- DOI: 10.7150/ijbs.3.455
Adjuvant effect of anti-4-1BB mAb administration in adoptive T cell therapy of cancer
Abstract
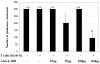
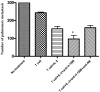
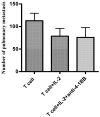
Administration of anti-4-1BB mAb has been found to be a potent adjuvant when combined with other therapeutic approaches, e.g. chemotherapy, cytokine therapies, anti-OX40 therapy, and peptide or DC vaccines. However, the adjuvant effect of anti-4-1BB mAb administration in adoptive T cell therapy of cancer has not been fully evaluated. In this report, effector T cells were generated in vitro by anti-CD3/anti-CD28 activation of tumor-draining lymph node (TDLN) cells and used in an adoptive immunotherapy model. While T cells or anti-4-1BB alone showed no therapeutic efficacy in mice bearing macroscopic 10-day pulmonary metastases, T cells plus anti-4-1BB mediated significant tumor regression in an anti-4-1BB dose dependent manner. Mice bearing microscopic 3-day lung metastases treated with T cells alone demonstrated tumor regression which was significantly enhanced by anti-4-1BB administration. NK cell depletion abrogated the augmented therapeutic efficacy rendered by anti-4-1BB. Cell transfer between congenic hosts demonstrated that anti-4-1BB administration increased the survival of adoptively transferred TDLN cells. Using STAT4(-/-) mice, we found that modulated IFN gamma secretion in wt TDLN cells after anti-CD3/CD28/4-1BB activation in vitro was lost in similarly stimulated STAT4(-/-) TDLN cells. Additionally, anti-4-1BB administration failed to augment the therapeutic efficacy of T cell therapy in STAT4(-/-) mice. Together, these results indicate that administered anti-4-1BB mAb can serve as an effective adjuvant to augment the antitumor reactivity of adoptively transferred T cells by recruiting the host NK cells; increasing the persistence of infused effector T cells, and modulating the STAT4 molecular signaling pathway.
Keywords: Adoptive immunotherapy; Anti-4-1BB; Cancer; NK cells; STAT4; T cells.
Conflict of interest statement
Conflict of Interests: The authors have declared that no conflict of interest exists.
Figures


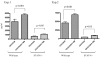
References
-
- Chang AE, Li Q, Jiang G et al. Phase II trial of autologous tumor vaccination, Anti-CD3-activated vaccine-primed lymphocytes, and Interleukin-2 in stage IV renal cell cancer. J Clin Oncol. 2003;21:884–890. - PubMed
-
- Topalian SL, Solomon D, Avis FP et al. Immunotherapy of patients with advanced cancer using tumor-infiltrating lymphocytes and recombinant interleukin-2: a pilot study. J Clin Oncol. 1988;6:839–853. - PubMed
-
- Ganss R, Ryschich E, Klar E et al. Combination of T-cell therapy and trigger of inflammation induces remodeling of the vasculature and tumor eradication. Cancer Res. 2002;62:1462–1470. - PubMed
-
- Huang H, Li F, Gordon JR et al. Synergistic enhancement of antitumor immunity with adoptively transferred tumor-specific CD4+ and CD8+ T cells and intratumoral lymphotactin transgene expression. Cancer Res. 2002;62:2043–2051. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

