Biphasic incorporation of centromeric histone CENP-A in fission yeast
- PMID: 18077559
- PMCID: PMC2230595
- DOI: 10.1091/mbc.e07-05-0504
Biphasic incorporation of centromeric histone CENP-A in fission yeast
Abstract
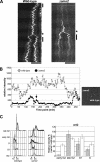
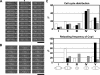
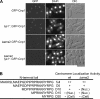
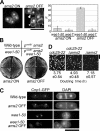
CENP-A is a centromere-specific histone H3 variant that is essential for kinetochore formation. Here, we report that the fission yeast Schizosaccharomyces pombe has at least two distinct CENP-A deposition phases across the cell cycle: S and G2. The S phase deposition requires Ams2 GATA factor, which promotes histone gene activation. In Delta ams2, CENP-A fails to retain during S, but it reaccumulates onto centromeres via the G2 deposition pathway, which is down-regulated by Hip1, a homologue of HIRA histone chaperon. Reducing the length of G2 in Delta ams2 results in failure of CENP-A accumulation, leading to chromosome missegregation. N-terminal green fluorescent protein-tagging reduces the centromeric association of CENP-A, causing cell death in Delta ams2 but not in wild-type cells, suggesting that the N-terminal tail of CENP-A may play a pivotal role in the formation of centromeric nucleosomes at G2. These observations imply that CENP-A is normally localized to centromeres in S phase in an Ams2-dependent manner and that the G2 pathway may salvage CENP-A assembly to promote genome stability. The flexibility of CENP-A incorporation during the cell cycle may account for the plasticity of kinetochore formation when the authentic centromere is damaged.
Figures

References
-
- Ahmad K., Henikoff S. The histone variant H3.3 marks active chromatin by replication-independent nucleosome assembly. Mol. Cell. 2002;9:1191–1200. - PubMed
-
- Black B. E., Foltz D. R., Chakravarthy S., Luger K., Woods V. L., Jr, Cleveland D. W. Structural determinants for generating centromeric chromatin. Nature. 2004;430:578–582. - PubMed
-
- Black B. E., Jansen L. E., Maddox P. S., Foltz D. R., Desai A. B., Shah J. V., Cleveland D. W. Centromere identity maintained by nucleosomes assembled with histone H3 containing the CENP-A targeting domain. Mol. Cell. 2007;25:309–322. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

