Involvement of MAPK signaling molecules and Runx2 in the NELL1-induced osteoblastic differentiation
- PMID: 18082140
- PMCID: PMC2959102
- DOI: 10.1016/j.febslet.2007.12.006
Involvement of MAPK signaling molecules and Runx2 in the NELL1-induced osteoblastic differentiation
Abstract
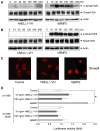
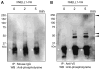
NELL1 is an extracellular protein inducing osteogenic differentiation and bone formation of osteoblastic cells. To elucidate the intracellular signaling cascade evoked by NELL1, we have shown that NELL1 protein transiently activates the MAPK signaling cascade, induces the phosphorylation of Runx2, and promotes the rapid intracellular accumulation of Tyr-phosphorylated proteins. Unlike BMP2, NELL1 protein does not activate the Smad signaling cascade. These findings suggest that upon binding to a specific receptor NELL1 transduces an osteogenic signal through activation of certain Tyr-kinases associated with the Ras-MAPK cascade, and finally leads to the osteogenic differentiation.
Figures




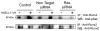
References
-
- Hughes FJ, Turner W, Belibasakis G, Martuscelli G. Effects of growth factors and cytokines on osteoblast differentiation. Periodontology. 2000;41:48–72. - PubMed
-
- Canalis E, Economides AN, Gazzerro E. Bone morphogenetic proteins, their antagonists, and the skeleton. Endocrinology. 2003;24:218–235. - PubMed
-
- Chen D, Zhao M, Mundy GR. Bone morphogenetic proteins. Growth Factors. 2004;22:233–241. - PubMed
-
- Ting K, Vastardis H, Mulliken JB, Soo C, Tieu A, Do H, Kwong E, Bertolami CN, Kawamoto H, Kuroda S, Longaker MT. Human NELL-1 expressed in unilateral coronal synostosis. J Bone Miner Res. 1999;14:80–89. - PubMed
-
- Kuroda S, Oyasu M, Kawakami M, Kanayama N, Tanizawa K, Saito N, Abe T, Matsuhashi S, Ting K. Biochemical characterization and expression analysis of neural thrombospondin-1-like proteins NELL1 and NELL2. Biochem Biophys Res Commun. 1999;265:79–86. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

