Intracellular interleukin-1alpha mediates interleukin-8 production induced by Chlamydia trachomatis infection via a mechanism independent of type I interleukin-1 receptor
- PMID: 18086816
- PMCID: PMC2258806
- DOI: 10.1128/IAI.01313-07
Intracellular interleukin-1alpha mediates interleukin-8 production induced by Chlamydia trachomatis infection via a mechanism independent of type I interleukin-1 receptor
Abstract
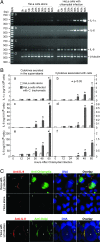
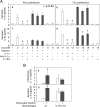
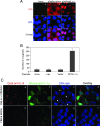
Chlamydia trachomatis infection induces a wide array of inflammatory cytokines and chemokines, which may contribute to chlamydia-induced pathologies. However, the precise mechanisms by which Chlamydia induces cytokines remain unclear. Here we demonstrate that the proinflammatory cytokine interleukin-1alpha (IL-1alpha) plays an essential role in chlamydial induction of the chemokine IL-8. Cells deficient in IL-1alpha expression or IL-1alpha-competent cells treated with IL-1alpha-specific small interfering RNA failed to produce IL-8 in response to chlamydial infection. However, neutralization of extracellular IL-1alpha or blockade of or deficiency in type I IL-1 receptor (IL-1RI) signaling did not affect chlamydial induction of IL-8 in cells capable of producing IL-1alpha. These results suggest that IL-1alpha can mediate the chlamydial induction of IL-8 via an intracellular mechanism independent of IL-1RI, especially during the early stage of the infection cycle. This conclusion is further supported by the observations that expression of a transgene-encoded full-length IL-1alpha fusion protein in the nuclei enhanced IL-8 production and that nuclear localization of chlamydia-induced precursor IL-1alpha correlated with chlamydial induction of IL-8. Thus, we have identified a novel mechanism for chlamydial induction of the chemokine IL-8.
Figures

Similar articles
-
Chlamydia trachomatis infection results in a modest pro-inflammatory cytokine response and a decrease in T cell chemokine secretion in human polarized endocervical epithelial cells.Cytokine. 2013 Aug;63(2):151-65. doi: 10.1016/j.cyto.2013.04.022. Epub 2013 May 11. Cytokine. 2013. PMID: 23673287 Free PMC article.
-
Secretion of proinflammatory cytokines by epithelial cells in response to Chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis.J Clin Invest. 1997 Jan 1;99(1):77-87. doi: 10.1172/JCI119136. J Clin Invest. 1997. PMID: 9011579 Free PMC article.
-
Interaction of Chlamydia trachomatis serovar E with male genital tract epithelium results in secretion of proinflammatory cytokines.J Med Microbiol. 2007 Aug;56(Pt 8):1025-1032. doi: 10.1099/jmm.0.47241-0. J Med Microbiol. 2007. PMID: 17644708
-
The cytosolic pattern recognition receptor NOD1 induces inflammatory interleukin-8 during Chlamydia trachomatis infection.Infect Immun. 2008 Jul;76(7):3150-5. doi: 10.1128/IAI.00104-08. Epub 2008 Apr 21. Infect Immun. 2008. PMID: 18426885 Free PMC article.
-
Role of cytokines in Chlamydia trachomatis protective immunity and immunopathology.Curr Pharm Des. 2003;9(1):67-73. doi: 10.2174/1381612033392486. Curr Pharm Des. 2003. PMID: 12570676 Review.
Cited by
-
Redox-control of the alarmin, Interleukin-1α.Redox Biol. 2013 Apr 17;1(1):218-25. doi: 10.1016/j.redox.2013.03.001. eCollection 2013. Redox Biol. 2013. PMID: 24024155 Free PMC article.
-
CRISPR/Cas9 mediated mutation of mouse IL-1α nuclear localisation sequence abolishes expression.Sci Rep. 2017 Dec 6;7(1):17077. doi: 10.1038/s41598-017-17387-x. Sci Rep. 2017. PMID: 29213066 Free PMC article.
-
Killing me softly: chlamydial use of proteolysis for evading host defenses.Trends Microbiol. 2009 Oct;17(10):467-74. doi: 10.1016/j.tim.2009.07.007. Epub 2009 Sep 16. Trends Microbiol. 2009. PMID: 19765998 Free PMC article. Review.
-
Interleukin 1 alpha administration is neuroprotective and neuro-restorative following experimental ischemic stroke.J Neuroinflammation. 2019 Nov 14;16(1):222. doi: 10.1186/s12974-019-1599-9. J Neuroinflammation. 2019. PMID: 31727174 Free PMC article.
-
Chlamydiaphage φCPG1 Capsid Protein Vp1 Inhibits Chlamydia trachomatis Growth via the Mitogen-Activated Protein Kinase Pathway.Viruses. 2016 Apr 14;8(4):99. doi: 10.3390/v8040099. Viruses. 2016. PMID: 27089359 Free PMC article.
References
-
- Banda, N. K., C. Guthridge, D. Sheppard, K. S. Cairns, M. Muggli, D. Bech-Otschir, W. Dubiel, and W. P. Arend. 2005. Intracellular IL-1 receptor antagonist type 1 inhibits IL-1-induced cytokine production in keratinocytes through binding to the third component of the COP9 signalosome. J. Immunol. 1743608-3616. - PubMed
-
- Bando, M., Y. Hiroshima, M. Kataoka, Y. Shinohara, M. C. Herzberg, K. F. Ross, T. Nagata, and J. I. Kido. 2007. Interleukin-1alpha regulates antimicrobial peptide expression in human keratinocytes. Immunol. Cell Biol. 85532-537. - PubMed
-
- Bauwens, J. E., H. Orlander, M. P. Gomez, M. Lampe, S. Morse, W. E. Stamm, R. Cone, R. Ashley, P. Swenson, and K. K. Holmes. 2002. Epidemic lymphogranuloma venereum during epidemics of crack cocaine use and HIV infection in the Bahamas. Sex. Transm. Dis. 29253-259. - PubMed
-
- Boraschi, D., and A. Tagliabue. 2006. The interleukin-1 receptor family. Vitam. Horm. 74229-254. - PubMed
-
- Buchholz, K. R., and R. S. Stephens. 2006. Activation of the host cell proinflammatory interleukin-8 response by Chlamydia trachomatis. Cell. Microbiol. 81768-1779. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

