Identification of a novel population of Langerin+ dendritic cells
- PMID: 18086865
- PMCID: PMC2150989
- DOI: 10.1084/jem.20071966
Identification of a novel population of Langerin+ dendritic cells
Abstract
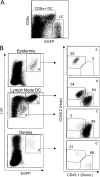
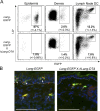
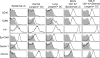
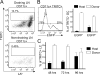
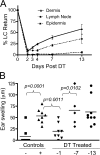
Langerhans cells (LCs) are antigen-presenting cells that reside in the epidermis of the skin and traffic to lymph nodes (LNs). The general role of these cells in skin immune responses is not clear because distinct models of LC depletion resulted in opposite conclusions about their role in contact hypersensitivity (CHS) responses. While comparing these models, we discovered a novel population of LCs that resides in the dermis and does not represent migrating epidermal LCs, as previously thought. Unlike epidermal LCs, dermal Langerin(+) dendritic cells (DCs) were radiosensitive and displayed a distinct cell surface phenotype. Dermal Langerin(+) DCs migrate from the skin to the LNs after inflammation and in the steady state, and represent the majority of Langerin(+) DCs in skin draining LNs. Both epidermal and dermal Langerin(+) DCs were depleted by treatment with diphtheria toxin in Lang-DTREGFP knock-in mice. In contrast, transgenic hLang-DTA mice lack epidermal LCs, but have normal numbers of dermal Langerin(+) DCs. CHS responses were abrogated upon depletion of both epidermal and dermal LCs, but were unaffected in the absence of only epidermal LCs. This suggests that dermal LCs can mediate CHS and provides an explanation for previous differences observed in the two-model systems.
Figures

References
-
- Stoitzner, P., K. Pfaller, H. Stossel, and N. Romani. 2002. A close-up view of migrating Langerhans cells in the skin. J. Invest. Dermatol. 118:117–125. - PubMed
-
- Stoitzner, P., S. Holzmann, A.D. McLellan, L. Ivarsson, H. Stossel, M. Kapp, U. Kammerer, P. Douillard, E. Kampgen, F. Koch, et al. 2003. Visualization and characterization of migratory Langerhans cells in murine skin and lymph nodes by antibodies against Langerin/CD207. J. Invest. Dermatol. 120:266–274. - PubMed
-
- Mayerova, D., E.A. Parke, L.S. Bursch, O.A. Odumade, and K.A. Hogquist. 2004. Langerhans cells activate naive self-antigen-specific CD8 T cells in the steady state. Immunity. 21:391–400. - PubMed
-
- Valladeau, J., O. Ravel, C. Dezutter-Dambuyant, K. Moore, M. Kleijmeer, Y. Liu, V. Duvert-Frances, C. Vincent, D. Schmitt, J. Davoust, et al. 2000. Langerin, a novel C-type lectin specific to Langerhans cells, is an endocytic receptor that induces the formation of Birbeck granules. Immunity. 12:71–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

