Nck adaptor proteins control the organization of neuronal circuits important for walking
- PMID: 18093944
- PMCID: PMC2409251
- DOI: 10.1073/pnas.0710316105
Nck adaptor proteins control the organization of neuronal circuits important for walking
Abstract
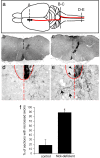
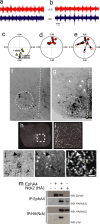
The intracellular signaling targets used by mammalian axon guidance receptors to organize the nervous system in vivo are unclear. The Nck1 and Nck2 SH2/SH3 adaptors (collectively Nck) can couple phosphotyrosine (pTyr) signals to reorganization of the actin cytoskeleton and are therefore candidates for linking guidance cues to the regulatory machinery of the cytoskeleton. We find that selective inactivation of Nck in the murine nervous system causes a hopping gait and a defect in the spinal central pattern generator, which is characterized by synchronous firing of bilateral ventral motor neurons. Nck-deficient mice also show abnormal projections of corticospinal tract axons and defective development of the posterior tract of the anterior commissure. These phenotypes are consistent with a role for Nck in signaling initiated by different classes of guidance receptors, including the EphA4 receptor tyrosine kinase. Our data indicate that Nck adaptors couple pTyr guidance signals to cytoskeletal events required for the ipsilateral projections of spinal cord neurons and thus for normal limb movement.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Jones N, Blasutig IM, Eremina V, Ruston JM, Bladt F, Li H, Huang H, Larose L, Li SS, Takano T, et al. Nature. 2006;440:818–823. - PubMed
-
- Li W, Fan J, Woodley DT. Oncogene. 2001;20:6403–6417. - PubMed
-
- Stein E, Huynh-Do U, Lane AA, Cerretti DP, Daniel TO. J Biol Chem. 1998;273:1303–1308. - PubMed
-
- Cowan CA, Henkemeyer M. Nature. 2001;413:174–179. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

