Contributions of a disulfide bond to the structure, stability, and dimerization of human IgG1 antibody CH3 domain
- PMID: 18156469
- PMCID: PMC2144599
- DOI: 10.1110/ps.073134408
Contributions of a disulfide bond to the structure, stability, and dimerization of human IgG1 antibody CH3 domain
Abstract
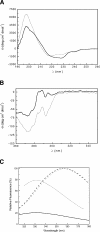
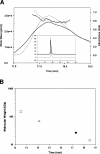
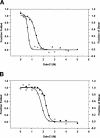
Recombinant human monoclonal antibodies have become important protein-based therapeutics for the treatment of various diseases. The antibody structure is complex, consisting of beta-sheet rich domains stabilized by multiple disulfide bridges. The dimerization of the C(H)3 domain in the constant region of the heavy chain plays a pivotal role in the assembly of an antibody. This domain contains a single buried, highly conserved disulfide bond. This disulfide bond was not required for dimerization, since a recombinant human C(H)3 domain, even in the reduced state, existed as a dimer. Spectroscopic analyses showed that the secondary and tertiary structures of reduced and oxidized C(H)3 dimer were similar, but differences were observed. The reduced C(H)3 dimer was less stable than the oxidized form to denaturation by guanidinium chloride (GdmCl), pH, or heat. Equilibrium sedimentation revealed that the reduced dimer dissociated at lower GdmCl concentration than the oxidized form. This implies that the disulfide bond shifts the monomer-dimer equilibrium. Interestingly, the dimer-monomer dissociation transition occurred at lower GdmCl concentration than the unfolding transition. Thus, disulfide bond formation in the human C(H)3 domain is important for stability and dimerization. Here we show the importance of the role played by the disulfide bond and how it affects the stability and monomer-dimer equilibrium of the human C(H)3 domain. Hence, these results may have implications for the stability of the intact antibody.
Figures




References
-
- Arakawa, T., Tsumoto, K. The effects of arginine on refolding of aggregated proteins: Not facilitate refolding, but suppress aggregation. Biochem. Biophys. Res. Commun. 2003;304:148–152. - PubMed
-
- Ashikari, Y., Arata, Y., Hamaguchi, K. pH-induced unfolding of the constant fragment of the immunoglobulin light chain: Effect of reduction of the intrachain disulfide bond. J. Biochem. 1985;97:517–528. - PubMed
-
- Azuma, H., Hayashi, T., Dent, J.A., Ruggeri, Z.M., Ware, J. Disulfide bond requirements for assembly of the platelet glycoprotein Ib-binding domain of von Willebrand factor. J. Biol. Chem. 1993;268:2821–2827. - PubMed
-
- Becker, G.W., Hsiung, H.M. Expression, secretion and folding of human growth hormone in Escherichia coli. Purification and characterization. FEBS Lett. 1986;204:145–150. - PubMed
-
- Benson, A.M., Suruda, A.J., Talalay, P. Concentration-dependent association of Δ5-3-ketosteroid isomerase of Pseudomonas testosteroni . J. Biol. Chem. 1975;250:276–280. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
