N-(4-Hydroxyphenyl)retinamide induced differentiation with repression of telomerase and cell cycle to increase interferon-gamma sensitivity for apoptosis in human glioblastoma cells
- PMID: 18164543
- PMCID: PMC3103940
- DOI: 10.1016/j.canlet.2007.11.016
N-(4-Hydroxyphenyl)retinamide induced differentiation with repression of telomerase and cell cycle to increase interferon-gamma sensitivity for apoptosis in human glioblastoma cells
Abstract
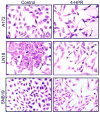
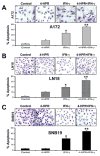
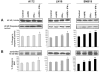
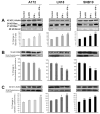
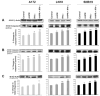
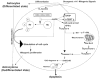
Glioblastoma is the most malignant and prevalent brain tumor in humans. It is composed of heterogenic abnormal astroglial cells that avoid differentiation, maintain proliferation, and hardly commit apoptosis. N-(4-Hydroxyphenyl)retinamide (4-HPR) induced astrocytic differentiation and increased sensitivity to interferon-gamma (IFN-gamma) for apoptosis in human glioblastoma A172, LN18, and SNB19 cells. Combination of 4-HPR and IFN-gamma significantly inhibited human telomerase reverse transcriptase (hTERT), cyclin dependent kinase 2 (CDK2), and survivin to up-regulate caspase-8, caspase-9, and caspase-3 for increasing apoptosis in all glioblastoma cell lines. Hence, combination of 4-HPR and IFN-gamma should be considered for controlling growth of different human glioblastoma cells.
Figures

Similar articles
-
N-(4-Hydroxyphenyl) retinamide potentiated paclitaxel for cell cycle arrest and apoptosis in glioblastoma C6 and RG2 cells.Brain Res. 2009 May 1;1268:142-153. doi: 10.1016/j.brainres.2009.02.064. Epub 2009 Mar 10. Brain Res. 2009. PMID: 19285047 Free PMC article.
-
N-(4-Hydroxyphenyl) retinamide induced both differentiation and apoptosis in human glioblastoma T98G and U87MG cells.Brain Res. 2008 Aug 28;1227:207-15. doi: 10.1016/j.brainres.2008.06.045. Epub 2008 Jun 21. Brain Res. 2008. PMID: 18602901 Free PMC article.
-
Molecular mechanisms of the combination of retinoid and interferon-gamma for inducing differentiation and increasing apoptosis in human glioblastoma T98G and U87MG cells.Neurochem Res. 2009 Jan;34(1):87-101. doi: 10.1007/s11064-008-9669-x. Epub 2008 Mar 27. Neurochem Res. 2009. PMID: 18368485 Free PMC article.
-
Combination of all-trans retinoic acid and interferon-gamma upregulated p27(kip1) and down regulated CDK2 to cause cell cycle arrest leading to differentiation and apoptosis in human glioblastoma LN18 (PTEN-proficient) and U87MG (PTEN-deficient) cells.Cancer Chemother Pharmacol. 2008 Aug;62(3):407-16. doi: 10.1007/s00280-007-0619-0. Epub 2007 Oct 25. Cancer Chemother Pharmacol. 2008. PMID: 17960384 Free PMC article.
-
Differentiation decreased telomerase activity in rat glioblastoma C6 cells and increased sensitivity to IFN-gamma and taxol for apoptosis.Neurochem Res. 2007 Dec;32(12):2167-83. doi: 10.1007/s11064-007-9413-y. Epub 2007 Aug 13. Neurochem Res. 2007. PMID: 17694433
Cited by
-
Fenretinide combines perturbation of signaling kinases, cell-extracellular matrix interactions and matrix metalloproteinase activation to inhibit invasion in oral squamous cell carcinoma cells.Carcinogenesis. 2022 Oct 22;43(9):851-864. doi: 10.1093/carcin/bgac070. Carcinogenesis. 2022. PMID: 35974187 Free PMC article.
-
N-(4-Hydroxyphenyl) retinamide potentiated paclitaxel for cell cycle arrest and apoptosis in glioblastoma C6 and RG2 cells.Brain Res. 2009 May 1;1268:142-153. doi: 10.1016/j.brainres.2009.02.064. Epub 2009 Mar 10. Brain Res. 2009. PMID: 19285047 Free PMC article.
-
Knockdown of hTERT and concurrent treatment with interferon-gamma inhibited proliferation and invasion of human glioblastoma cell lines.Int J Biochem Cell Biol. 2010 Jul;42(7):1164-73. doi: 10.1016/j.biocel.2010.04.002. Epub 2010 Apr 13. Int J Biochem Cell Biol. 2010. PMID: 20394835 Free PMC article.
-
N-Myc down regulation induced differentiation, early cell cycle exit, and apoptosis in human malignant neuroblastoma cells having wild type or mutant p53.Biochem Pharmacol. 2009 Nov 1;78(9):1105-14. doi: 10.1016/j.bcp.2009.06.009. Epub 2009 Jun 18. Biochem Pharmacol. 2009. PMID: 19540207 Free PMC article.
-
High levels of β-catenin promote IFNγ-induced apoptosis in hepatocellular carcinoma cells.Oncol Lett. 2012 Nov;4(5):1092-1096. doi: 10.3892/ol.2012.844. Epub 2012 Aug 6. Oncol Lett. 2012. PMID: 23162658 Free PMC article.
References
-
- Ray SK, Patel SJ, Welsh CT, Wilford GG, Hogan EL, Banik NL. Molecular evidence of apoptotic death in malignant brain tumors including glioblastoma multiforme: upregulation of calpain and caspase-3. J Neurosci Res. 2002;69:197–206. - PubMed
-
- Kleihues P, Louis DN, Scheithauer BW, Rorke LB, Reifenberger G, Burger PC, et al. The WHO classification of tumors of the nervous system. J Neuropathol Exp Neurol. 2002;61:215–225. - PubMed
-
- Parney IF, Chang SM. Current chemotherapy for glioblastoma. Cancer J. 2003;9:149–156. - PubMed
-
- Parney IF, Hao C, Petruk KC. Glioblastoma immunology and immunotherapy. Neurosurgery. 2000;46:778–91. - PubMed
-
- Chambon PA. Decade of molecular biology of retinoic acid receptors. FASEB J. 1996;10:940–954. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

