Molecular basis of synaptic vesicle cargo recognition by the endocytic sorting adaptor stonin 2
- PMID: 18166656
- PMCID: PMC2373488
- DOI: 10.1083/jcb.200708107
Molecular basis of synaptic vesicle cargo recognition by the endocytic sorting adaptor stonin 2
Abstract
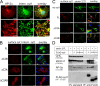
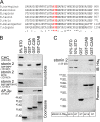
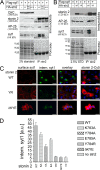
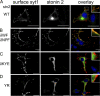
Synaptic transmission depends on clathrin-mediated recycling of synaptic vesicles (SVs). How select SV proteins are targeted for internalization has remained elusive. Stonins are evolutionarily conserved adaptors dedicated to endocytic sorting of the SV protein synaptotagmin. Our data identify the molecular determinants for recognition of synaptotagmin by stonin 2 or its Caenorhabditis elegans orthologue UNC-41B. The interaction involves the direct association of clusters of basic residues on the surface of the cytoplasmic domain of synaptotagmin 1 and a beta strand within the mu-homology domain of stonin 2. Mutation of K783, Y784, and E785 to alanine within this stonin 2 beta strand results in failure of the mutant stonin protein to associate with synaptotagmin, to accumulate at synapses, and to facilitate synaptotagmin internalization. Synaptotagmin-binding-defective UNC-41B is unable to rescue paralysis in C. elegans stonin mutant animals, suggesting that the mechanism of stonin-mediated SV cargo recognition is conserved from worms to mammals.
Figures


Similar articles
-
Stonins--specialized adaptors for synaptic vesicle recycling and beyond?Traffic. 2010 Jan;11(1):8-15. doi: 10.1111/j.1600-0854.2009.00971.x. Traffic. 2010. PMID: 19732400 Review.
-
Stonin 2 is an AP-2-dependent endocytic sorting adaptor for synaptotagmin internalization and recycling.Dev Cell. 2006 Feb;10(2):233-44. doi: 10.1016/j.devcel.2005.12.011. Dev Cell. 2006. PMID: 16459302
-
UNC-41/stonin functions with AP2 to recycle synaptic vesicles in Caenorhabditis elegans.PLoS One. 2012;7(7):e40095. doi: 10.1371/journal.pone.0040095. Epub 2012 Jul 10. PLoS One. 2012. PMID: 22808098 Free PMC article.
-
Compromised fidelity of endocytic synaptic vesicle protein sorting in the absence of stonin 2.Proc Natl Acad Sci U S A. 2013 Feb 5;110(6):E526-35. doi: 10.1073/pnas.1218432110. Epub 2013 Jan 23. Proc Natl Acad Sci U S A. 2013. PMID: 23345427 Free PMC article.
-
Signals involved in targeting membrane proteins to synaptic vesicles.Cell Mol Neurobiol. 2002 Dec;22(5-6):565-77. doi: 10.1023/a:1021884319363. Cell Mol Neurobiol. 2002. PMID: 12585680 Free PMC article. Review.
Cited by
-
A distributed set of interactions controls mu2 functionality in the role of AP-2 as a sorting adaptor in synaptic vesicle endocytosis.J Biol Chem. 2009 Nov 20;284(47):32803-12. doi: 10.1074/jbc.M109.039149. Epub 2009 Sep 17. J Biol Chem. 2009. PMID: 19762466 Free PMC article.
-
Safeguards of Neurotransmission: Endocytic Adaptors as Regulators of Synaptic Vesicle Composition and Function.Front Cell Neurosci. 2017 Oct 11;11:320. doi: 10.3389/fncel.2017.00320. eCollection 2017. Front Cell Neurosci. 2017. PMID: 29085282 Free PMC article. Review.
-
Exploring the eukaryotic Yip and REEP/Yop superfamily of membrane-shaping adapter proteins (MSAPs): A cacophony or harmony of structure and function?Front Mol Biosci. 2022 Aug 19;9:912848. doi: 10.3389/fmolb.2022.912848. eCollection 2022. Front Mol Biosci. 2022. PMID: 36060263 Free PMC article. Review.
-
AP2 hemicomplexes contribute independently to synaptic vesicle endocytosis.Elife. 2013 Mar 5;2:e00190. doi: 10.7554/eLife.00190. Elife. 2013. PMID: 23482940 Free PMC article.
-
SNT-1 Functions as the Ca2+ Sensor for Tonic and Evoked Neurotransmitter Release in Caenorhabditis Elegans.J Neurosci. 2018 Jun 6;38(23):5313-5324. doi: 10.1523/JNEUROSCI.3097-17.2018. Epub 2018 May 14. J Neurosci. 2018. PMID: 29760174 Free PMC article.
References
-
- Bai, J., W.C. Tucker, and E.R. Chapman. 2004. PIP2 increases the speed of response of synaptotagmin and steers its membrane-penetration activity toward the plasma membrane. Nat. Struct. Mol. Biol. 11:36–44. - PubMed
-
- Bonifacino, J.S., and L.M. Traub. 2003. Signals for sorting of transmembrane proteins to endosomes and lysosomes. Annu. Rev. Biochem. 72:395–447. - PubMed
-
- Brodin, L., P. Low, and O. Shupliakov. 2000. Sequential steps in clathrin-mediated synaptic vesicle endocytosis. Curr. Opin. Neurobiol. 10:312–320. - PubMed
-
- Chapman, E.R., R.C. Desai, A.F. Davis, and C.K. Tornehl. 1998. Delineation of the oligomerization, AP-2 binding, and synprint binding region of the C2B domain of synaptotagmin. J. Biol. Chem. 273:32966–32972. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

