Dioxin-mediated tumor progression through activation of mitochondria-to-nucleus stress signaling
- PMID: 18172213
- PMCID: PMC2224183
- DOI: 10.1073/pnas.0706183104
Dioxin-mediated tumor progression through activation of mitochondria-to-nucleus stress signaling
Abstract
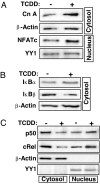
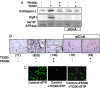
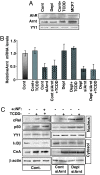
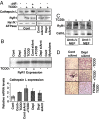
The environmental toxin 2,3,7,8-tetrachlorodibenzodioxin (TCDD) is a known human carcinogen; however, its precise mechanism of action remains unclear. Here we show that TCDD induces mitochondrial dysfunction, stress signaling, and tumor invasion by a mechanism similar to that described for mtDNA-depleted cells. Treatment of C2C12 cells with TCDD disrupted mitochondrial transmembrane potential in a time-dependent fashion and inhibited mitochondrial transcription and translation. TCDD also increased cytosolic [Ca(2+)](c) and RyR1-specific Ca(2+) release. These changes were associated with increased calcineurin (CnA) levels and activation of CnA-sensitive NF-kappaB/Rel (IkappaBbeta-dependent) factors. Cells treated with TCDD displayed resistance to apoptosis, increased expression of the tumor marker cathepsin L, and a high degree of invasiveness as tested by the Matrigel membrane invasion assay. These effects were reversed by the CnA inhibitor FK506, and CnA mRNA silencing suggesting that TCDD triggers a signaling pathway similar to mtDNA depletion. Taken together, these results reveal that TCDD may promote tumor progression in vivo by directly targeting mitochondrial transcription and induction of mitochondrial stress signaling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

