Transrepression function of the glucocorticoid receptor regulates eyelid development and keratinocyte proliferation but is not sufficient to prevent skin chronic inflammation
- PMID: 18174358
- PMCID: PMC5419553
- DOI: 10.1210/me.2007-0284
Transrepression function of the glucocorticoid receptor regulates eyelid development and keratinocyte proliferation but is not sufficient to prevent skin chronic inflammation
Abstract
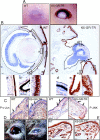
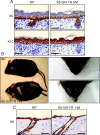
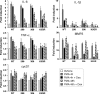
Glucocorticoids (GCs) play a key role in skin homeostasis and stress responses acting through the GC receptor (GR), which modulates gene expression by DNA binding-dependent (transactivation) and -independent (transrepression) mechanisms. To delineate which mechanisms underlie the beneficial and adverse effects mediated by GR in epidermis and other epithelia, we have generated transgenic mice that express a mutant GR (P493R, A494S), which is defective for transactivation but retains transrepression activity, under control of the keratin 5 promoter (K5-GR-TR mice). K5-GR-TR embryos exhibited eyelid opening at birth and corneal defects that resulted in corneal opacity in the adulthood. Transgenic embryos developed normal skin, although epidermal atrophy and focal alopecia was detected in adult mice. GR-mediated transrepression was sufficient to inhibit keratinocyte proliferation induced by acute and chronic phorbol 12-myristate 13-acetate exposure, as demonstrated by morphometric analyses, bromodeoxyuridine incorporation, and repression of keratin 6, a marker of hyperproliferative epidermis. These antiproliferative effects were mediated through negative interference of GR with MAPK/activator protein-1 and nuclear factor-kappaB activities, although these interactions occurred with different kinetics. However, phorbol 12-myristate 13-acetate-induced inflammation was only partially inhibited by GR-TR, which efficiently repressed IL-1beta and MMP-3 genes while weakly repressing IL-6 and TNF-alpha. Our data highlight the relevance of deciphering the mechanisms underlying GR actions on epithelial morphogenesis as well as for its therapeutic use to identify more restricted targets of GC administration.
Figures




References
-
- Rhen T, Cidlowski JA 2005. Antiinflammatory action of glucocorticoids: new mechanisms for old drugs. N Engl J Med 353:1711–1723 - PubMed
-
- Cole TJ, Blendy AP, Monaghan K, Schmid W, Aguzzi A, Fantuzzi G, Hummler E, Unsicker K, Schütz G 1995. Targeted disruption of the glucocorticoid receptor gene blocks adrenergic chromaffin cell development and severely retards lung maturation. Genes Dev 9:1608–1621 - PubMed
-
- Buttgereit F, Burmester G, Lipworth BJ 2005. Optimised glucocorticoid therapy: the sharpening of an old spear. Lancet 365:801–803 - PubMed
-
- Seckl JR, Nyirenda MJ, Walker BR, Chapman KE 1999. Glucocorticoids and fetal programming. Biochem Soc Trans 27:74–78 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

