TLR ligands that stimulate the metabolism of vitamin D3 in activated murine dendritic cells can function as effective mucosal adjuvants to subcutaneously administered vaccines
- PMID: 18178294
- PMCID: PMC2632860
- DOI: 10.1016/j.vaccine.2007.11.084
TLR ligands that stimulate the metabolism of vitamin D3 in activated murine dendritic cells can function as effective mucosal adjuvants to subcutaneously administered vaccines
Abstract
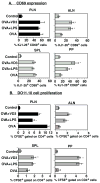
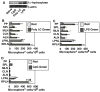
Cathelicidin production by human myeloid cells stimulated through toll-like receptor (TLR) 2/1, the migration of human CD8+ T cells to inflamed skin sites, and the ability of murine dendritic cells (DCs) to migrate from skin sites of vaccination to mucosal lymphoid organs all occur via calcitriol-dependent mechanisms. Herein, we report that murine DCs exposed to TLR3/TLR4 ligands upregulate their expression of 1 alpha-hydroxylase, the enzyme that converts circulating 25(OH)D3 to calcitriol, the active form of vitamin D3. TLR3/TLR4 ligands injected subcutaneously affect DC migration in vivo, allowing their trafficking to both draining and non-draining systemic and mucosal lymphoid organs. Subcutaneously delivered vaccines containing TLR3/TLR4 ligands and antigen stimulate the induction of both systemic and mucosal immune responses. Vaccines containing TLR9 ligands fail to stimulate 1 alpha-hydroxylase protein expression, are incapable of redirecting DC migration into Peyer's patches and do not induce mucosal immune responses. These findings support a hypothesis that active metabolites of vitamin D3 produced locally are able to affect various aspects of innate and acquired immune responses.
Figures



Similar articles
-
TLR-induced local metabolism of vitamin D3 plays an important role in the diversification of adaptive immune responses.J Immunol. 2009 Apr 1;182(7):4296-305. doi: 10.4049/jimmunol.0804344. J Immunol. 2009. PMID: 19299729
-
Vitamin D3-mediated alterations to myeloid dendritic cell trafficking in vivo expand the scope of their antigen presenting properties.Vaccine. 2007 Jan 26;25(7):1236-49. doi: 10.1016/j.vaccine.2006.10.008. Epub 2006 Oct 24. Vaccine. 2007. PMID: 17092617
-
Toll-like receptor 2 suppresses Toll-like receptor 9 responses in Peyer's patch dendritic cells.Immunobiology. 2015 Jun;220(6):734-43. doi: 10.1016/j.imbio.2014.12.022. Epub 2015 Jan 8. Immunobiology. 2015. PMID: 25638261
-
Peyer's patch dendritic cells as regulators of mucosal adaptive immunity.Cell Mol Life Sci. 2005 Jun;62(12):1333-8. doi: 10.1007/s00018-005-5037-z. Cell Mol Life Sci. 2005. PMID: 15971108 Free PMC article. Review.
-
Manipulating systemic and mucosal immune responses with skin-deliverable adjuvants.J Biotechnol. 1996 Jan 26;44(1-3):13-9. doi: 10.1016/0168-1656(95)00099-2. J Biotechnol. 1996. PMID: 8717381 Review.
Cited by
-
Can we translate vitamin D immunomodulating effect on innate and adaptive immunity to vaccine response?Nutrients. 2015 Mar 20;7(3):2044-60. doi: 10.3390/nu7032044. Nutrients. 2015. PMID: 25803545 Free PMC article. Review.
-
Regulation of Dendritic Cell Function by Vitamin D.Nutrients. 2015 Sep 21;7(9):8127-51. doi: 10.3390/nu7095383. Nutrients. 2015. PMID: 26402698 Free PMC article. Review.
-
Therapeutic DNA vaccine induces broad T cell responses in the gut and sustained protection from viral rebound and AIDS in SIV-infected rhesus macaques.PLoS One. 2012;7(3):e33715. doi: 10.1371/journal.pone.0033715. Epub 2012 Mar 19. PLoS One. 2012. PMID: 22442716 Free PMC article.
-
The Safety and Immunogenicity of the mRNA-BNT162b2 SARS-CoV-2 Vaccine in Hemodialysis Patients.Front Immunol. 2021 Jun 16;12:704773. doi: 10.3389/fimmu.2021.704773. eCollection 2021. Front Immunol. 2021. PMID: 34220867 Free PMC article.
-
Environmental cues, dendritic cells and the programming of tissue-selective lymphocyte trafficking.Nat Immunol. 2008 Sep;9(9):981-7. doi: 10.1038/ni.f.208. Nat Immunol. 2008. PMID: 18711435 Free PMC article. Review.
References
-
- Takeda K, Kaisho T, Akira S. Toll-like receptors. Annu Rev Immunol. 2003;21:335–76. - PubMed
-
- Smiley ST, King JA, Hancock WW. Fibrinogen stimulates macrophage chemokine secretion through toll-like receptor 4. J Immunol. 2001;167(5):2887–94. - PubMed
-
- Su SL, Tsai CD, Lee CH, Salter DM, Lee HS. Expression and regulation of Toll-like receptor 2 by IL-1beta and fibronectin fragments in human articular chondrocytes. Osteoarthritis Cartilage. 2005;13(10):879–86. - PubMed
-
- Biragyn A, Ruffini PA, Leifer CA, Klyushnenkova E, Shakhov A, Chertov O, et al. Toll-like receptor 4-dependent activation of dendritic cells by beta-defensin 2. Science. 2002;298(5595):1025–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

