In vivo MRI identifies cholinergic circuitry deficits in a Down syndrome model
- PMID: 18180075
- PMCID: PMC2771203
- DOI: 10.1016/j.neurobiolaging.2007.11.026
In vivo MRI identifies cholinergic circuitry deficits in a Down syndrome model
Abstract
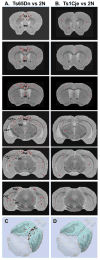
In vivo quantitative magnetic resonance imaging (MRI) was employed to detect brain pathology and map its distribution within control, disomic mice (2N) and in Ts65Dn and Ts1Cje trisomy mice with features of human Down syndrome (DS). In Ts65Dn, but not Ts1Cje mice, transverse proton spin-spin (T(2)) relaxation time was selectively reduced in the medial septal nucleus (MSN) and in brain regions that receive cholinergic innervation from the MSN, including the hippocampus, cingulate cortex, and retrosplenial cortex. Basal forebrain cholinergic neurons (BFCNs) in the MSN, identified by choline acetyltransferase (ChAT) and nerve growth factor receptors p75(NTR) and TrkA immunolabeling were reduced in Ts65Dn brains and in situ acetylcholinesterase (AChE) activity was depleted distally along projecting cholinergic fibers, and selectively on pre- and postsynaptic profiles in these target areas. T(2) effects were negligible in Ts1Cje mice that are diploid for App and lack BFCN neuropathology, consistent with the suspected relationship of this pathology to increased App dosage. These results establish the utility of quantitative MRI in vivo for identifying Alzheimer's disease-relevant cholinergic changes in animal models of DS and characterizing the selective vulnerability of cholinergic neuron subpopulations.
Conflict of interest statement
Disclosure Statement: The authors have no actual or potential conflicts of interest to disclose.
Figures





References
-
- Antonarakis SE, Lyle R, Chrast R, Scott HS. Differential gene expression studies to explore the molecular pathophysiology of Down syndrome. Brain Res Brain Res Rev. 2001;36(23):265–74. - PubMed
-
- Antonarakis SE, Lyle R, Dermitzakis ET, Reymond A, Deutsch S. Chromosome 21 and down syndrome: from genomics to pathophysiology. Nat Rev Genet. 2004;5(10):725–38. - PubMed
-
- Apelt J, Kumar A, Schliebs R. Impairment of cholinergic neurotransmission in adult and aged transgenic Tg2576 mouse brain expressing the Swedish mutation of human beta-amyloid precursor protein. Brain Res. 2002;953(12):17–30. - PubMed
-
- Ardekani BA, Guckemus S, Bachman A, Hoptman MJ, Wojtaszek M, Nierenberg J. Quantitative comparison of algorithms for inter-subject registration of 3D volumetric brain MRI scans. J Neurosci Methods. 2005;142(1):67–76. - PubMed
-
- Bakshi R, Benedict RH, Bermel RA, Caruthers SD, Puli SR, Tjoa CW, Fabiano AJ, Jacobs L. T2 hypointensity in the deep gray matter of patients with multiple sclerosis: a quantitative magnetic resonance imaging study. Arch Neurol. 2002;59(1):62–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

