Freeze-dried tendon allografts as tissue-engineering scaffolds for Gdf5 gene delivery
- PMID: 18180771
- PMCID: PMC2705193
- DOI: 10.1038/sj.mt.6300395
Freeze-dried tendon allografts as tissue-engineering scaffolds for Gdf5 gene delivery
Abstract
Tendon reconstruction using grafts often results in adhesions that limit joint flexion. These adhesions are precipitated by inflammation, fibrosis, and the paucity of tendon differentiation signals during healing. In order to study this problem, we developed a mouse model in which the flexor digitorum longus (FDL) tendon is reconstructed using a live autograft or a freeze-dried allograft, and identified growth and differentiation factor 5 (Gdf5) as a therapeutic target. In this study we have investigated the potential of rAAV-Gdf5 -loaded freeze-dried tendon allografts as "therapeutically endowed" tissue-engineering scaffolds to reduce adhesions. In reporter gene studies we have demonstrated that recombinant adeno-associated virus (rAAV)-loaded tendon allografts mediate efficient transduction of adjacent soft tissues, with expression peaking at 7 days. We have also demonstrated that the rAAV-Gdf5 vector significantly accelerates wound healing in an in vitro fibroblast scratch model and, when loaded onto freeze-dried FDL tendon allografts, improves the metatarsophalangeal (MTP) joint flexion to a significantly greater extent than the rAAV-lacZ controls do. Collectively, our data demonstrate the feasibility and efficacy of therapeutic tendon allograft processing as a novel paradigm in tissue engineering in order to address difficult clinical problems such as tendon adhesions.
Figures

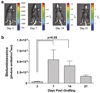

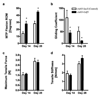

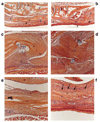
References
-
- Lilly SI, Messer TM. Complications after treatment of flexor tendon injuries. J Am Acad Orthop Surg. 2006;14:387–396. - PubMed
-
- Gelberman RH, Manske PR. Factors influencing flexor tendon adhesions. Hand Clin. 1985;1:35–42. - PubMed
-
- Silva MJ, Boyer MI, Gelberman RH. Recent progress in flexor tendon healing. J Orthop Sci. 2002;7:508–514. - PubMed
-
- Leddy JP. Flexor Tendons - Acute Injuries. In: Green DP, editor. Operative Hand Surgery. Vol. 3. New York: Churchill Livingstone; 1988. pp. 1935–1968.
-
- Whitlock PW, Smith TL, Poehling GG, Shilt JS, Van Dyke M. A naturally derived, cytocompatible, and architecturally optimized scaffold for tendon and ligament regeneration. Biomaterials. 2007;28:4321–4329. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

