Heat-shock protein 90alpha1 is required for organized myofibril assembly in skeletal muscles of zebrafish embryos
- PMID: 18182494
- PMCID: PMC2206574
- DOI: 10.1073/pnas.0707330105
Heat-shock protein 90alpha1 is required for organized myofibril assembly in skeletal muscles of zebrafish embryos
Abstract
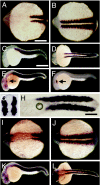
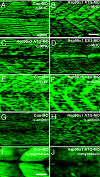
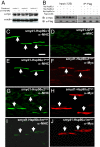
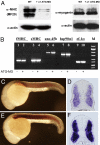
Heat-shock protein 90alpha (Hsp90alpha) is a member of the molecular chaperone family involved in protein folding and assembly. The role of Hsp90alpha in the developmental process, however, remains unclear. Here we report that zebrafish contains two Hsp90alpha genes, Hsp90alpha1, and Hsp90alpha2. Hsp90alpha1 is specifically expressed in developing somites and skeletal muscles of zebrafish embryos. We have demonstrated that Hsp90alpha1 is essential for myofibril organization in skeletal muscles of zebrafish embryos. Knockdown of Hsp90alpha1 resulted in paralyzed zebrafish embryos with poorly organized myofibrils in skeletal muscles. In contrast, knockdown of Hsp90alpha2 had no effect on muscle contraction and myofibril organization. The filament defects could be rescued in a cell autonomous manner by an ectopic expression of Hsp90alpha1. Biochemical analyses revealed that knockdown of Hsp90alpha1 resulted in significant myosin degradation and up-regulation of unc-45b gene expression. These results indicate that Hsp90alpha1 plays an important role in muscle development, likely through facilitating myosin folding and assembly into organized myofibril filaments.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Knockdown and overexpression of Unc-45b result in defective myofibril organization in skeletal muscles of zebrafish embryos.BMC Cell Biol. 2010 Sep 17;11:70. doi: 10.1186/1471-2121-11-70. BMC Cell Biol. 2010. PMID: 20849610 Free PMC article.
-
The Effects of Hsp90α1 Mutations on Myosin Thick Filament Organization.PLoS One. 2015 Nov 12;10(11):e0142573. doi: 10.1371/journal.pone.0142573. eCollection 2015. PLoS One. 2015. PMID: 26562659 Free PMC article.
-
Loss of Smyhc1 or Hsp90alpha1 function results in different effects on myofibril organization in skeletal muscles of zebrafish embryos.PLoS One. 2010 Jan 1;5(1):e8416. doi: 10.1371/journal.pone.0008416. PLoS One. 2010. PMID: 20049323 Free PMC article.
-
UCS proteins: chaperones for myosin and co-chaperones for Hsp90.Subcell Biochem. 2015;78:133-52. doi: 10.1007/978-3-319-11731-7_7. Subcell Biochem. 2015. PMID: 25487020 Review.
-
Assembly and Maintenance of Myofibrils in Striated Muscle.Handb Exp Pharmacol. 2017;235:39-75. doi: 10.1007/164_2016_53. Handb Exp Pharmacol. 2017. PMID: 27832381 Review.
Cited by
-
Cloning, molecular characterization, and expression analysis of the unc45 myosin chaperone b(unc45b)gene of grass carp (Ctenopharyngodon idellus).J Muscle Res Cell Motil. 2016 Jun;37(3):71-81. doi: 10.1007/s10974-016-9445-5. Epub 2016 Jun 22. J Muscle Res Cell Motil. 2016. PMID: 27334505
-
Generation of MuRF-GFP transgenic zebrafish models for investigating murf gene expression and protein localization in Smyd1b and Hsp90α1 knockdown embryos.Comp Biochem Physiol B Biochem Mol Biol. 2020 Feb;240:110368. doi: 10.1016/j.cbpb.2019.110368. Epub 2019 Oct 24. Comp Biochem Physiol B Biochem Mol Biol. 2020. PMID: 31669374 Free PMC article.
-
UCS Chaperone Folding of the Myosin Head: A Function That Evolved before Animals and Fungi Diverged from a Common Ancestor More than a Billion Years Ago.Biomolecules. 2022 Jul 26;12(8):1028. doi: 10.3390/biom12081028. Biomolecules. 2022. PMID: 35892339 Free PMC article. Review.
-
Zebrafish Embryonic Slow Muscle Is a Rapid System for Genetic Analysis of Sarcomere Organization by CRISPR/Cas9, but Not NgAgo.Mar Biotechnol (NY). 2018 Apr;20(2):168-181. doi: 10.1007/s10126-018-9794-8. Epub 2018 Jan 27. Mar Biotechnol (NY). 2018. PMID: 29374849
-
The HSP90 chaperone machinery.Nat Rev Mol Cell Biol. 2017 Jun;18(6):345-360. doi: 10.1038/nrm.2017.20. Epub 2017 Apr 21. Nat Rev Mol Cell Biol. 2017. PMID: 28429788 Review.
References
-
- Clark KA, McElhinny AS, Beckerle MC, Gregorio CC. Annu Rev Cell Dev Biol. 2002;18:637–706. - PubMed
-
- Laing NG, Nowak KJ. BioEssays. 2005;27:809–822. - PubMed
-
- Hutagalung AH, Landsverk ML, Price MG, Epstein HF. J Cell Sci. 2002;115:3983–3990. - PubMed
-
- Epstein HF, Thomson JN. Nature. 1974;250:579–580. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

