Probing the role of parasite-specific, distant structural regions on communication and catalysis in the bifunctional thymidylate synthase-dihydrofolate reductase from Plasmodium falciparum
- PMID: 18189414
- PMCID: PMC2658879
- DOI: 10.1021/bi701624u
Probing the role of parasite-specific, distant structural regions on communication and catalysis in the bifunctional thymidylate synthase-dihydrofolate reductase from Plasmodium falciparum
Abstract
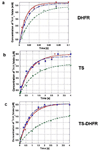
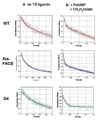
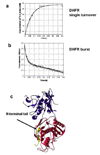
Plasmodium falciparum thymidylate synthase-dihydrofolate reductase (TS-DHFR) is an essential enzyme in nucleotide biosynthesis and a validated molecular drug target in malaria. Because P. falciparum TS and DHFR are highly homologous to their human counterparts, existing active-site antifolate drugs can have dose-limiting toxicities. In humans, TS and DHFR are two separate proteins. In P. falciparum, however, TS-DHFR is bifunctional, with both TS and DHFR active sites on a single polypeptide chain of the enzyme. Consequently, P. falciparum TS-DHFR contains unique distant or nonactive regions that might modulate catalysis: (1) an N-terminal tail and (2) a linker region tethering DHFR to TS, and encoding a crossover helix that forms critical electrostatic interactions with the DHFR active site. The role of these nonactive sites in the bifunctional P. falciparum TS-DHFR is unknown. We report the first in-depth, pre-steady-state kinetic characterization of the full-length, wild-type (WT) P. falciparum TS-DHFR enzyme and probe the role of distant, nonactive regions through mutational analysis. We show that the overall rate-limiting step in the WT P. falciparum TS-DHFR enzyme is TS catalysis. We further show that if TS is in an activated (liganded) conformation, the DHFR rate is 2-fold activated, from 60 s-1 to 130 s-1 in the WT enzyme. The TS rate is also reciprocally activated by approximately 1.5-fold if DHFR is in an activated, ligand-bound conformation. Mutations to the linker region affect neither catalytic rate nor domain-domain communication. Deletion of the N-terminal tail, although in a location remote from the active site, decreases the DHFR single rate and the bifunctional TS-DHFR rate by a factor of 2. The 2-fold activation of the DHFR rate by TS ligands remains intact, although even the activated N-terminal mutant has just half the DHFR activity of the WT enzyme. However, the reciprocal communication between TS active site and DHFR ligands is impaired in N-terminal mutants. Surprisingly, deletion of the analogous N-terminal tail in Leishmania major TS-DHFR causes a 3-fold enhancement of the DHFR rate from approximately 14 s-1 to approximately 40 s-1. In summary, our results demonstrate a complex interplay of domain-domain communication and nonactive-site modulation of catalysis in P. falciparum TS-DHFR. Furthermore, each parasitic TS-DHFR is activated by unique mechanisms, modulated by their nonactive site regions. Finally, our studies suggest the N-terminal tail of P. falciparum TS-DHFR is a highly selective, novel target for potential antifolate development in malaria.
Figures


Similar articles
-
Understanding the molecular mechanism of substrate channeling and domain communication in protozoal bifunctional TS-DHFR.Protein Eng Des Sel. 2017 Mar 1;30(3):253-261. doi: 10.1093/protein/gzx004. Protein Eng Des Sel. 2017. PMID: 28338744 Free PMC article. Review.
-
First three-dimensional structure of Toxoplasma gondii thymidylate synthase-dihydrofolate reductase: insights for catalysis, interdomain interactions, and substrate channeling.Biochemistry. 2013 Oct 15;52(41):7305-7317. doi: 10.1021/bi400576t. Epub 2013 Oct 3. Biochemistry. 2013. PMID: 24053355 Free PMC article.
-
Disruption of the crossover helix impairs dihydrofolate reductase activity in the bifunctional enzyme TS-DHFR from Cryptosporidium hominis.Biochem J. 2009 Feb 1;417(3):757-64. doi: 10.1042/BJ20081247. Biochem J. 2009. PMID: 18851711 Free PMC article.
-
Kinetic reaction scheme for the dihydrofolate reductase domain of the bifunctional thymidylate synthase-dihydrofolate reductase from Leishmania major.Biochemistry. 1998 Sep 1;37(35):12206-12. doi: 10.1021/bi9803170. Biochemistry. 1998. PMID: 9724534
-
Thymidylate synthase-dihydrofolate reductase in protozoa.Exp Parasitol. 1990 Apr;70(3):367-71. doi: 10.1016/0014-4894(90)90119-w. Exp Parasitol. 1990. PMID: 2178951 Review.
Cited by
-
Understanding the molecular mechanism of substrate channeling and domain communication in protozoal bifunctional TS-DHFR.Protein Eng Des Sel. 2017 Mar 1;30(3):253-261. doi: 10.1093/protein/gzx004. Protein Eng Des Sel. 2017. PMID: 28338744 Free PMC article. Review.
-
Multiple origins and regional dispersal of resistant dhps in African Plasmodium falciparum malaria.PLoS Med. 2009 Apr 14;6(4):e1000055. doi: 10.1371/journal.pmed.1000055. Epub 2009 Apr 14. PLoS Med. 2009. PMID: 19365539 Free PMC article.
-
Purine and pyrimidine pathways as targets in Plasmodium falciparum.Curr Top Med Chem. 2011;11(16):2103-15. doi: 10.2174/156802611796575948. Curr Top Med Chem. 2011. PMID: 21619511 Free PMC article. Review.
-
Insights into the role of the junctional region of Plasmodium falciparum dihydrofolate reductase-thymidylate synthase.Malar J. 2013 Mar 12;12:91. doi: 10.1186/1475-2875-12-91. Malar J. 2013. PMID: 23497065 Free PMC article.
-
Interactions between cycloguanil derivatives and wild type and resistance-associated mutant Plasmodium falciparum dihydrofolate reductases.J Comput Aided Mol Des. 2009 Apr;23(4):241-52. doi: 10.1007/s10822-008-9254-z. Epub 2009 Jan 21. J Comput Aided Mol Des. 2009. PMID: 19156529
References
-
- Greenwood BM, et al. Malaria. Lancet. 2005;365(9469):1487–1498. - PubMed
-
- Ivanetich KM, Santi DV. Thymidylate synthase-dihydrofolate reductase in protozoa. Exp Parasitol. 1990;70(3):367–371. - PubMed
-
- Yuthavong Y. Basis for antifolate action and resistance in malaria. Microbes Infect. 2002;4(2):175–182. - PubMed
-
- Ivanetich KM, Santi DV. Bifunctional thymidylate synthase-dihydrofolate reductase in protozoa. Faseb J. 1990;4(6):1591–1597. - PubMed
-
- Atreya CE, Anderson KS. Kinetic characterization of bifunctional thymidylate synthasedihydrofolate reductase (TS-DHFR) from Cryptosporidium hominis: a paradigm shift for ts activity and channeling behavior. J Biol Chem. 2004;279(18):18314–18322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

