An HIV-1 clade C DNA prime, NYVAC boost vaccine regimen induces reliable, polyfunctional, and long-lasting T cell responses
- PMID: 18195071
- PMCID: PMC2234371
- DOI: 10.1084/jem.20071331
An HIV-1 clade C DNA prime, NYVAC boost vaccine regimen induces reliable, polyfunctional, and long-lasting T cell responses
Abstract
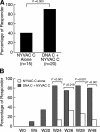
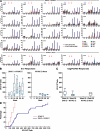
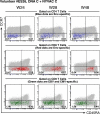
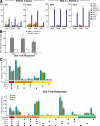
The EuroVacc 02 phase I trial has evaluated the safety and immunogenicity of a prime-boost regimen comprising recombinant DNA and the poxvirus vector NYVAC, both expressing a common immunogen consisting of Env, Gag, Pol, and Nef polypeptide domain from human immunodeficiency virus (HIV)-1 clade C isolate, CN54. 40 volunteers were randomized to receive DNA C or nothing on day 0 and at week 4, followed by NYVAC C at weeks 20 and 24. The primary immunogenicity endpoints were measured at weeks 26 and 28 by the quantification of T cell responses using the interferon gamma enzyme-linked immunospot assay. Our results indicate that the DNA C plus NYVAC C vaccine regimen was highly immunogenic, as indicated by the detection of T cell responses in 90% of vaccinees and was superior to responses induced by NYVAC C alone (33% of responders). The vaccine-induced T cell responses were (a) vigorous in the case of the env response (mean 480 spot-forming units/10(6) mononuclear cells at weeks 26/28), (b) polyfunctional for both CD4 and CD8 T cell responses, (c) broad (the average number of epitopes was 4.2 per responder), and (d) durable (T cell responses were present in 70% of vaccinees at week 72). The vaccine-induced T cell responses were strongest and most frequently directed against Env (91% of vaccines), but smaller responses against Gag-Pol-Nef were also observed in 48% of vaccinees. These results support the development of the poxvirus platform in the HIV vaccine field and the further clinical development of the DNA C plus NYVAC C vaccine regimen.
Figures




Comment in
-
The failed HIV Merck vaccine study: a step back or a launching point for future vaccine development?J Exp Med. 2008 Jan 21;205(1):7-12. doi: 10.1084/jem.20072681. Epub 2008 Jan 14. J Exp Med. 2008. PMID: 18195078 Free PMC article.
References
-
- Schwartländer, B., J. Stover, N. Walker, L. Bollinger, J.P. Gutierrez, W. McGreevey, M. Opuni, S. Forsythe, L. Kumaranayake, C. Watts, and S. Bertozzi. 2001. AIDS. Resource needs for HIV/AIDS. Science. 292:2434–2436. - PubMed
-
- Bart, P.A., A. Harari, and G. Pantaleo. 2006. Clinical studies of experimental vaccines. Current Opinion in HIV & AIDS. 1:286–293. - PubMed
-
- Letvin, N.L. 2006. Progress and obstacles in the development of an AIDS vaccine. Nat. Rev. Immunol. 6:930–939. - PubMed
-
- McMichael, A.J. 2006. HIV vaccines. Annu. Rev. Immunol. 24:227–255. - PubMed
-
- Heeney, J.L. 2004. Requirement of diverse T-helper responses elicited by HIV vaccines: induction of highly targeted humoral and CTL responses. Expert Rev. Vaccines. 3:S53–S64. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

