Antiapoptotic and trophic effects of dominant-negative forms of dual leucine zipper kinase in dopamine neurons of the substantia nigra in vivo
- PMID: 18199767
- PMCID: PMC2896687
- DOI: 10.1523/JNEUROSCI.2132-07.2008
Antiapoptotic and trophic effects of dominant-negative forms of dual leucine zipper kinase in dopamine neurons of the substantia nigra in vivo
Abstract
There is extensive evidence that the mitogen-activated protein kinase (MAPK) signaling cascade mediates programmed cell death in neurons. However, current evidence that the mixed linage kinases (MLKs), upstream in this cascade, mediate cell death is based, in the in vivo context, entirely on pharmacological approaches. The compounds used in these studies have neither complete specificity nor selectivity among these kinases. Therefore, to better address the molecular specificity of the MLKs in mediating neuron death, we used dominant-negative constructs delivered by AAV (adenoassociated virus) vector transfer. We assessed effects in a neurotoxin model of parkinsonism, in which neuroprotection by pharmacologic MLK inhibition has been reported. We find that two dominant-negative forms of dual leucine zipper kinase (DLK) inhibit apoptosis and enhance long-term survival of dopamine neurons, but a dominant negative of MLK3 does not. Interestingly, the kinase-dead form of DLK not only blocks apoptosis but also has trophic effects on dopamine neurons. Although the MAPK cascade activates a number of downstream cell death mediators, we find that inhibition of DLK correlates closely with blockade of phosphorylation of c-jun and prevention of cell death. We conclude that DLK acts primarily through c-jun phosphorylation to mediate cell death in this model.
Figures
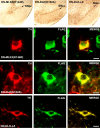
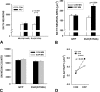
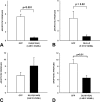

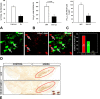
References
-
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. - PubMed
-
- Coleman M. Axon degeneration mechanisms: commonality amid diversity. Nat Rev Neurosci. 2005;6:889–898. - PubMed
-
- Donovan N, Becker EB, Konishi Y, Bonni A. JNK phosphorylation and activation of BAD couples the stress-activated signaling pathway to the cell death machinery. J Biol Chem. 2002;277:40944–40949. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
