Direct transfer of starter substrates from type I fatty acid synthase to type III polyketide synthases in phenolic lipid synthesis
- PMID: 18199837
- PMCID: PMC2242712
- DOI: 10.1073/pnas.0709819105
Direct transfer of starter substrates from type I fatty acid synthase to type III polyketide synthases in phenolic lipid synthesis
Abstract
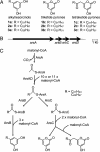
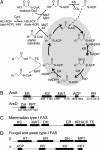
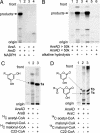
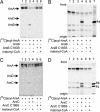
Alkylresorcinols and alkylpyrones, which have a polar aromatic ring and a hydrophobic alkyl chain, are phenolic lipids found in plants, fungi, and bacteria. In the Gram-negative bacterium Azotobacter vinelandii, phenolic lipids in the membrane of dormant cysts are essential for encystment. The aromatic moieties of the phenolic lipids in A. vinelandii are synthesized by two type III polyketide synthases (PKSs), ArsB and ArsC, which are encoded by the ars operon. However, details of the synthesis of hydrophobic acyl chains, which might serve as starter substrates for the type III polyketide synthases (PKSs), were unknown. Here, we show that two type I fatty acid synthases (FASs), ArsA and ArsD, which are members of the ars operon, are responsible for the biosynthesis of C(22)-C(26) fatty acids from malonyl-CoA. In vivo and in vitro reconstitution of phenolic lipid synthesis systems with the Ars enzymes suggested that the C(22)-C(26) fatty acids produced by ArsA and ArsD remained attached to the ACP domain of ArsA and were transferred hand-to-hand to the active-site cysteine residues of ArsB and ArsC. The type III PKSs then used the fatty acids as starter substrates and carried out two or three extensions with malonyl-CoA to yield the phenolic lipids. The phenolic lipids in A. vinelandii were thus found to be synthesized solely from malonyl-CoA by the four members of the ars operon. This is the first demonstration that a type I FAS interacts directly with a type III PKS through substrate transfer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

