Fission and selective fusion govern mitochondrial segregation and elimination by autophagy
- PMID: 18200046
- PMCID: PMC2234339
- DOI: 10.1038/sj.emboj.7601963
Fission and selective fusion govern mitochondrial segregation and elimination by autophagy
Abstract
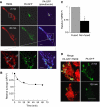
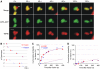
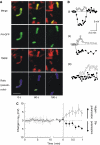
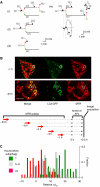
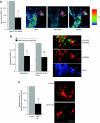
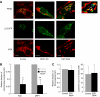
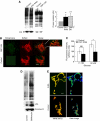
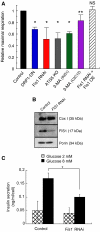
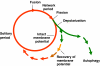
Accumulation of depolarized mitochondria within beta-cells has been associated with oxidative damage and development of diabetes. To determine the source and fate of depolarized mitochondria, individual mitochondria were photolabeled and tracked through fusion and fission. Mitochondria were found to go through frequent cycles of fusion and fission in a 'kiss and run' pattern. Fission events often generated uneven daughter units: one daughter exhibited increased membrane potential (delta psi(m)) and a high probability of subsequent fusion, while the other had decreased membrane potential and a reduced probability for a fusion event. Together, this pattern generated a subpopulation of non-fusing mitochondria that were found to have reduced delta psi(m) and decreased levels of the fusion protein OPA1. Inhibition of the fission machinery through DRP1(K38A) or FIS1 RNAi decreased mitochondrial autophagy and resulted in the accumulation of oxidized mitochondrial proteins, reduced respiration and impaired insulin secretion. Pulse chase and arrest of autophagy at the pre-proteolysis stage reveal that before autophagy mitochondria lose delta psi(m) and OPA1, and that overexpression of OPA1 decreases mitochondrial autophagy. Together, these findings suggest that fission followed by selective fusion segregates dysfunctional mitochondria and permits their removal by autophagy.
Figures
References
-
- Alavi MV, Bette S, Schimpf S, Schuettauf F, Schraermeyer U, Wehrl HF, Ruttiger L, Beck SC, Tonagel F, Pichler BJ, Knipper M, Peters T, Laufs J, Wissinger B (2007) A splice site mutation in the murine Opa1 gene features pathology of autosomal dominant optic atrophy. Brain 130: 1029–1042 - PubMed
-
- Barsoum MJ, Yuan H, Gerencser AA, Liot G, Kushnareva Y, Graber S, Kovacs I, Lee WD, Waggoner J, Cui J, White AD, Bossy B, Martinou JC, Youle RJ, Lipton SA, Ellisman MH, Perkins GA, Bossy-Wetzel E (2006) Nitric oxide-induced mitochondrial fission is regulated by dynamin-related GTPases in neurons. EMBO J 25: 3900–3911 - PMC - PubMed
-
- Chan DC (2006) Mitochondria: dynamic organelles in disease, aging, and development. Cell 125: 1241–1252 - PubMed
-
- Chen H, Chomyn A, Chan DC (2005) Disruption of fusion results in mitochondrial heterogeneity and dysfunction. J Biol Chem 280: 26185–26192 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

