The utility of affinity-tags for detection of a streptococcal protein from a variety of streptococcal species
- PMID: 18201786
- PMCID: PMC2435255
- DOI: 10.1016/j.mimet.2007.12.002
The utility of affinity-tags for detection of a streptococcal protein from a variety of streptococcal species
Abstract
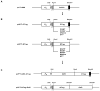
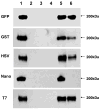
There is no systematic examination of affinity tag utility in Gram-positive bacteria, which limits the investigation of protein function in this important group of bacteria as specific antibodies for many of native proteins are generally not available. In this study, we utilized an E. coli-streptococcal shuttle vector pVT1666 and constructed two sets of expression plasmids pVPT-CTag and pVPT-NTag, with each set containing five affinity tags (GST, GFP, HSV, T7 and Nano) that can be fused to either the C- or N-terminus of a target protein. A putative glycosyltransferase (Gtf2) essential for Fap1 glycosylation was used to demonstrate the utility of the cassettes in detection of Gtf2 fusion proteins, and the biological relevance of the proteins in our working strain Streptococcus parasanguinis. GFP and T7 tags were readily expressed in S. parasanguinis as either an N- or C-terminal fusion to Gtf2. Only the C- terminal fusion of GST and HSV were able to be identified in S. parasanguinis. The Nano tag was not detected in either E. coli or S. parasanguinis. Genetic complementation experiments indicated that all the tagged Gtf2 fusion proteins could restore the Gtf2 function in the null mutant except for the Nano-tagged Gtf2 at its N-terminal fusion. Using a T7-tagged Gtf2 fusion construct, we demonstrated that the fusion cassette is also useful in detection of the fusion tag expression in other streptococci including S. mutans, S. pneumoniae and S. sanguinis. Therefore, the expression cassettes we constructed will be a useful tool not only to investigate protein-protein interactions in Fap1 biogenesis in S. parasanguinis, but also to study protein functions in other gram-positive bacteria in which pVT1666 replicates.
Figures


References
-
- Ajdiæ D, McShan WM, McLaughlin RE, Saviæ G, Chang J, Carson MB, Primeaux C, Tian R, Kenton S, Jia H, Lin S, Qian Y, Li S, Zhu H, Najar F, Lai H, White J, Roe BA, Ferretti JJ. Genome sequence of Streptococcus mutans UA159, a cariogenic dental pathogen. Proc Natl Acad Sci U S A. 2002;99:14434–9. - PMC - PubMed
-
- Arnau J, Lauritzen C, Petersen GE, Pedersen J. Current strategies for the use of affinity tags and tag removal for the purification of recombinant proteins. Protein Expr Purif. 2006;48:1–13. - PubMed
-
- Biedendieck R, Yang Y, Deckwer WD, Malten M, Jahn D. Plasmid system for the intracellular production and purification of affinity-tagged proteins in Bacillus megaterium. Biotechnol Bioeng. 2007;96:525–37. - PubMed
-
- Bricker AL, Camilli A. Transformation of a type 4 encapsulated strain of Streptococcus pneumoniae. FEMS Microbiol Lett. 1999;172:131–5. - PubMed
-
- Carlsson J, Grahnén H, Jonsson G, Wikner S. Establishment of Streptococcus sanguis in the mouths of infants. Arch Oral Biol. 1970;15:1143–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

