Amyloidogenic processing but not amyloid precursor protein (APP) intracellular C-terminal domain production requires a precisely oriented APP dimer assembled by transmembrane GXXXG motifs
- PMID: 18201969
- PMCID: PMC2702479
- DOI: 10.1074/jbc.M707142200
Amyloidogenic processing but not amyloid precursor protein (APP) intracellular C-terminal domain production requires a precisely oriented APP dimer assembled by transmembrane GXXXG motifs
Erratum in
- J Biol Chem. 2008 May 2;283(18):12680
Abstract
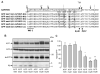
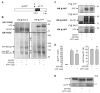
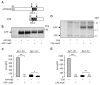
The beta-amyloid peptide (Abeta) is the major constituent of the amyloid core of senile plaques found in the brain of patients with Alzheimer disease. Abeta is produced by the sequential cleavage of the amyloid precursor protein (APP) by beta- and gamma-secretases. Cleavage of APP by gamma-secretase also generates the APP intracellular C-terminal domain (AICD) peptide, which might be involved in regulation of gene transcription. APP contains three Gly-XXX-Gly (GXXXG) motifs in its juxtamembrane and transmembrane (TM) regions. Such motifs are known to promote dimerization via close apposition of TM sequences. We demonstrate that pairwise replacement of glycines by leucines or isoleucines, but not alanines, in a GXXXG motif led to a drastic reduction of Abeta40 and Abeta42 secretion. beta-Cleavage of mutant APP was not inhibited, and reduction of Abeta secretion resulted from inhibition of gamma-cleavage. It was anticipated that decreased gamma-cleavage of mutant APP would result from inhibition of its dimerization. Surprisingly, mutations of the GXXXG motif actually enhanced dimerization of the APP C-terminal fragments, possibly via a different TM alpha-helical interface. Increased dimerization of the TM APP C-terminal domain did not affect AICD production.
Figures






Similar articles
-
Alanine substitutions in the GXXXG motif alter C99 cleavage by γ-secretase but not its dimerization.J Neurochem. 2017 Mar;140(6):955-962. doi: 10.1111/jnc.13942. Epub 2017 Jan 30. J Neurochem. 2017. PMID: 28032911
-
Major carboxyl terminal fragments generated by γ-secretase processing of the Alzheimer amyloid precursor are 50 and 51 amino acids long.Am J Geriatr Psychiatry. 2013 May;21(5):474-83. doi: 10.1016/j.jagp.2013.02.009. Am J Geriatr Psychiatry. 2013. PMID: 23570890 Free PMC article.
-
Lysine 624 of the amyloid precursor protein (APP) is a critical determinant of amyloid β peptide length: support for a sequential model of γ-secretase intramembrane proteolysis and regulation by the amyloid β precursor protein (APP) juxtamembrane region.J Biol Chem. 2011 Nov 18;286(46):39804-12. doi: 10.1074/jbc.M111.274696. Epub 2011 Aug 25. J Biol Chem. 2011. PMID: 21868378 Free PMC article.
-
Cellular membrane fluidity in amyloid precursor protein processing.Mol Neurobiol. 2014 Aug;50(1):119-29. doi: 10.1007/s12035-014-8652-6. Epub 2014 Feb 20. Mol Neurobiol. 2014. PMID: 24553856 Free PMC article. Review.
-
Cellular mechanisms of beta-amyloid production and secretion.Proc Natl Acad Sci U S A. 1999 Sep 28;96(20):11049-53. doi: 10.1073/pnas.96.20.11049. Proc Natl Acad Sci U S A. 1999. PMID: 10500121 Free PMC article. Review.
Cited by
-
Cholesterol-dependent amyloid β production: space for multifarious interactions between amyloid precursor protein, secretases, and cholesterol.Cell Biosci. 2023 Sep 13;13(1):171. doi: 10.1186/s13578-023-01127-y. Cell Biosci. 2023. PMID: 37705117 Free PMC article. Review.
-
Neuroprotective secreted amyloid precursor protein acts by disrupting amyloid precursor protein dimers.J Biol Chem. 2009 May 29;284(22):15016-25. doi: 10.1074/jbc.M808755200. Epub 2009 Mar 31. J Biol Chem. 2009. PMID: 19336403 Free PMC article.
-
The Metalloprotease Meprin β Is an Alternative β-Secretase of APP.Front Mol Neurosci. 2017 Jan 5;9:159. doi: 10.3389/fnmol.2016.00159. eCollection 2016. Front Mol Neurosci. 2017. PMID: 28105004 Free PMC article. Review.
-
Structures of beta-amyloid peptide 1-40, 1-42, and 1-55-the 672-726 fragment of APP-in a membrane environment with implications for interactions with gamma-secretase.J Am Chem Soc. 2009 Dec 16;131(49):17843-52. doi: 10.1021/ja905457d. J Am Chem Soc. 2009. PMID: 19995075 Free PMC article.
-
Amyloid precursor protein controls cholesterol turnover needed for neuronal activity.EMBO Mol Med. 2013 Apr;5(4):608-25. doi: 10.1002/emmm.201202215. EMBO Mol Med. 2013. PMID: 23554170 Free PMC article.
References
-
- Seubert P, Vigo-Pelfrey C, Esch F, Lee M, Dovey H, Davis D, Sinha S, Schlossmacher M, Whaley J, Swindlehurst C. Nature. 1992;359:325–327. - PubMed
-
- Shoji M, Golde TE, Ghiso J, Cheung TT, Estus S, Shaffer LM, Cai XD, McKay DM, Tintner R, Frangione B. Science. 1992;258:126–129. - PubMed
-
- Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH, Multhaup G, Beyreuther K, Muller-Hill B. Nature. 1987;325:733–736. - PubMed
-
- Sinha S, Anderson JP, Barbour R, Basi GS, Caccavello R, Davis D, Doan M, Dovey HF, Frigon N, Hong J, Jacobson-Croak K, Jewett N, Keim P, Knops J, Lieberburg I, Power M, Tan H, Tatsuno G, Tung J, Schenk D, Seubert P, Suomensaari SM, Wang S, Walker D, John V. Nature. 1999;402:537–540. - PubMed
-
- Vassar R, Bennett BD, Babu-Khan S, Kahn S, Mendiaz EA, Denis P, Teplow DB, Ross S, Amarante P, Loeloff R, Luo Y, Fisher S, Fuller J, Edenson S, Lile J, Jarosinski MA, Biere AL, Curran E, Burgess T, Louis JC, Collins F, Treanor J, Rogers G, Citron M. Science. 1999;286:735–741. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

