Carboxypeptidase E cytoplasmic tail-driven vesicle transport is key for activity-dependent secretion of peptide hormones
- PMID: 18202146
- PMCID: PMC2276472
- DOI: 10.1210/me.2007-0473
Carboxypeptidase E cytoplasmic tail-driven vesicle transport is key for activity-dependent secretion of peptide hormones
Abstract
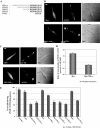
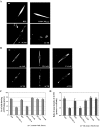
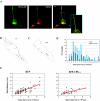
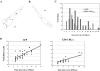
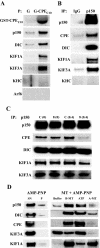
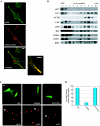
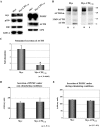
Vesicular transport of peptide hormones from the cell body to the plasma membrane for activity-dependent secretion is important for endocrine function, but how it is achieved is unclear. Here we uncover a mechanism in which the cytoplasmic tail of transmembrane carboxypeptidase E (CPE) found in proopiomelanocotin (POMC)/ACTH vesicles interacts with microtubule-based motors to control transport of these vesicles to the release site in pituitary cells. Overexpression of the CPE tail in live cells significantly reduced the velocity and distance of POMC/ACTH- and CPE-containing vesicle movement into the cell processes. Biochemical studies showed that the CPE tail interacted with dynactin, which, in turn, recruited microtubule plus-end motors kinesin 2 and kinesin 3. Overexpression of the CPE tail inhibited the stimulated secretion of ACTH from AtT20 cells. Thus, the CPE cytoplasmic tail interaction with dynactin-kinesin 2/kinesin 3 plays an important role in the transport of POMC vesicles for activity-dependent secretion.
Figures

Similar articles
-
A bi-directional carboxypeptidase E-driven transport mechanism controls BDNF vesicle homeostasis in hippocampal neurons.Mol Cell Neurosci. 2008 Sep;39(1):63-73. doi: 10.1016/j.mcn.2008.05.016. Epub 2008 Jun 4. Mol Cell Neurosci. 2008. PMID: 18573344 Free PMC article.
-
Carboxypeptidase E, Identified As a Direct Interactor of Growth Hormone, Is Important for Efficient Secretion of the Hormone.Mol Cells. 2016 Oct;39(10):756-761. doi: 10.14348/molcells.2016.0183. Epub 2016 Oct 28. Mol Cells. 2016. PMID: 27788574 Free PMC article.
-
Interaction between secretogranin III and carboxypeptidase E facilitates prohormone sorting within secretory granules.J Cell Sci. 2005 Oct 15;118(Pt 20):4785-95. doi: 10.1242/jcs.02608. J Cell Sci. 2005. PMID: 16219686
-
ACTH: cellular peptide hormone synthesis and secretory pathways.Results Probl Cell Differ. 2010;50:63-84. doi: 10.1007/400_2009_30. Results Probl Cell Differ. 2010. PMID: 19888563 Review.
-
Regulation of dynein-dynactin-driven vesicular transport.Traffic. 2017 Jun;18(6):336-347. doi: 10.1111/tra.12475. Epub 2017 Mar 28. Traffic. 2017. PMID: 28248450 Review.
Cited by
-
Going Too Far Is the Same as Falling Short†: Kinesin-3 Family Members in Hereditary Spastic Paraplegia.Front Cell Neurosci. 2019 Sep 26;13:419. doi: 10.3389/fncel.2019.00419. eCollection 2019. Front Cell Neurosci. 2019. PMID: 31616253 Free PMC article. Review.
-
Cthrc1, a novel circulating hormone regulating metabolism.PLoS One. 2012;7(10):e47142. doi: 10.1371/journal.pone.0047142. Epub 2012 Oct 8. PLoS One. 2012. PMID: 23056600 Free PMC article.
-
POMC: The Physiological Power of Hormone Processing.Physiol Rev. 2018 Oct 1;98(4):2381-2430. doi: 10.1152/physrev.00024.2017. Physiol Rev. 2018. PMID: 30156493 Free PMC article. Review.
-
Absence of carboxypeptidase E leads to adult hippocampal neuronal degeneration and memory deficits.Hippocampus. 2008;18(10):1051-63. doi: 10.1002/hipo.20462. Hippocampus. 2008. PMID: 18570185 Free PMC article.
-
Mini-GAGR, an intranasally applied polysaccharide, activates the neuronal Nrf2-mediated antioxidant defense system.J Biol Chem. 2018 Nov 23;293(47):18242-18269. doi: 10.1074/jbc.RA117.001245. Epub 2018 Oct 3. J Biol Chem. 2018. PMID: 30282635 Free PMC article.
References
-
- Hamm-Alvarez SF, Da Costa S, Yang T, Wei X, Gierow JP, Mircheff AK 1997 Cholinergic stimulation of lacrimal acinar cells promotes redistribution of membrane-associated kinesin and the secretory protein, β-hexosaminidase, and increases kinesin motor activity. Exp Eye Res 64:141–156 - PubMed
-
- Senda T, Yu W 1999 Kinesin cross-bridges between neurosecretory granules and microtubules in the mouse neurohypophysis. Neurosci Lett 262:69–71 - PubMed
-
- Pack-Chung E, Kurshan PT, Dickman DK, Schwarz TL 2007 A Drosophila kinesin required for synaptic bouton formation and synaptic vesicle transport. Nat Neurosci 10:980–989 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

