Distinct activities of the germline and somatic reproductive tissues in the regulation of Caenorhabditis elegans' longevity
- PMID: 18202391
- PMCID: PMC2206098
- DOI: 10.1534/genetics.107.083253
Distinct activities of the germline and somatic reproductive tissues in the regulation of Caenorhabditis elegans' longevity
Abstract
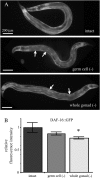
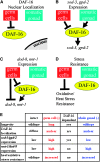
The two parts of the Caenorhabditis elegans reproductive system, the germ cells and the somatic reproductive tissues, each influence the life span of the animal. Removing the germ cells increases longevity, and this life span extension requires the somatic gonad. Here we show that the somatic gonad and the germ cells make distinct contributions to life span determination. The life span increase produced by loss of the germ cells requires the DAF-16/FOXO transcription factor. In response to germ-cell removal, DAF-16 accumulates in nuclei. We find that the somatic gonad is not required for DAF-16 nuclear accumulation or for the increased stress resistance that is produced by germ-cell removal. The somatic gonad is required, however, for expression of specific DAF-16 target genes. DAF-16 is known to be activated by reduced insulin/IGF-1 signaling in C. elegans. In certain insulin/IGF-1-pathway mutants, the somatic gonad is not required for germ-cell removal to extend life span. Our genetic experiments suggest that these mutations reduce insulin/IGF-1 signaling below a critical threshold level. At these low levels of insulin/IGF-1 signaling, factors normally provided by the somatic gonad are no longer needed for germ-cell removal to increase the expression of DAF-16 target genes.
Figures



References
-
- Arantes-Oliveira, N., J. Apfeld, A. Dillin and C. Kenyon, 2002. Regulation of life-span by germ-line stem cells in Caenorhabditis elegans. Science 295: 502–505. - PubMed
-
- Arantes-Oliveira, N., J. R. Berman and C. Kenyon, 2003. Healthy animals with extreme longevity. Science 302: 611. - PubMed
-
- Berman, J. R., and C. Kenyon, 2006. Germ-cell loss extends C. elegans life span through regulation of DAF-16 by kri-1 and lipophilic-hormone signaling. Cell 124: 1055–1068. - PubMed
-
- Broue, F., P. Liere, C. Kenyon and E. E. Baulieu, 2007. A steroid hormone that extends the lifespan of Caenorhabditis elegans. Aging Cell 6: 87–94. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

