Persistence of a chimerical phenotype after hepatocyte differentiation of human bone marrow mesenchymal stem cells
- PMID: 18211285
- PMCID: PMC6495711
- DOI: 10.1111/j.1365-2184.2007.00507.x
Persistence of a chimerical phenotype after hepatocyte differentiation of human bone marrow mesenchymal stem cells
Abstract
Objectives: Recent studies have suggested the potential of mesenchymal stem cells (MSCs) to differentiate into a hepatocyte-like lineage. Here, we evaluate the efficacy of hepatocyte differentiation of MSCs by studying acquisition of hepatocyte-like features together with alteration of the native mesenchymal phenotype.
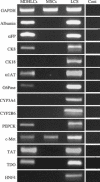
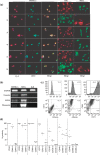
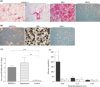
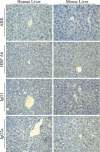
Material and methods: In vitro, we have investigated protein and mRNA level expression of hepatocyte and mesenchymal markers of mesenchymal-derived hepatocyte-like cells (MDHLCs) and we have evaluated their functionality using metabolic assays. In vivo, we investigated co-expression of hepatocyte (albumin, alpha-foetoprotein, cytokeratin 18) and mesenchymal (fibronectin, vimentin) markers after transplantation of MSCs or MDHLCs into severe combined immune deficiency mice.
Results: We observed that while in vitro these cells acquired some phenotypic and functional features of mature hepatocytes, they partially preserved their mesenchymal phenotype. After intrasplenic transplantation, engrafted MSCs with isolated expression of fibronectin and alpha-foetoprotein were observed. When these cells were injected into the liver, they expressed all analysed markers, confirming the chimaeric co-expression observed in vitro. Conversely, liver-engrafted MDHLCs conserved their hepatocyte-lineage markers but lost their chimaeric phenotype.
Conclusions: Hepatocyte differentiation of MSCs predominantly allows the acquisition of phenotypic hallmarks and provides chimaeric cells that maintain expression of initial lineage markers. However, advanced maturation to the hepatocyte-like phenotype could be obtained in vivo by conditioning MSCs prior to transplantation or by infusing cells into the liver micro-environment.
Figures





Similar articles
-
Differentiation of bone marrow-derived mesenchymal stem cells into hepatocyte-like cells on nanofibers and their transplantation into a carbon tetrachloride-induced liver fibrosis model.Stem Cell Rev Rep. 2011 Mar;7(1):103-18. doi: 10.1007/s12015-010-9126-5. Stem Cell Rev Rep. 2011. PMID: 20182823
-
Leukemia inhibitory factor contributes to hepatocyte-like differentiation of human bone marrow mesenchymal stem cells.Differentiation. 2008 Dec;76(10):1057-67. doi: 10.1111/j.1432-0436.2008.00287.x. Epub 2008 Jun 13. Differentiation. 2008. PMID: 18557762
-
Human skin fibroblasts: From mesodermal to hepatocyte-like differentiation.Hepatology. 2007 Nov;46(5):1574-85. doi: 10.1002/hep.21839. Hepatology. 2007. PMID: 17969047
-
Adipose tissue-derived mesenchymal stem cells and hepatic differentiation: old concepts and future perspectives.Eur Rev Med Pharmacol Sci. 2011 Apr;15(4):355-64. Eur Rev Med Pharmacol Sci. 2011. PMID: 21608430 Review.
-
Current status of human adipose-derived stem cells: differentiation into hepatocyte-like cells.ScientificWorldJournal. 2011;11:1568-81. doi: 10.1100/tsw.2011.146. Epub 2011 Aug 16. ScientificWorldJournal. 2011. PMID: 22224071 Free PMC article. Review.
Cited by
-
Stem cells for liver tissue repair: current knowledge and perspectives.World J Gastroenterol. 2008 Feb 14;14(6):864-75. doi: 10.3748/wjg.14.864. World J Gastroenterol. 2008. PMID: 18240343 Free PMC article. Review.
-
Mesenchymal stem cells as therapeutics and vehicles for gene and drug delivery.Adv Drug Deliv Rev. 2010 Sep 30;62(12):1156-66. doi: 10.1016/j.addr.2010.08.010. Epub 2010 Sep 7. Adv Drug Deliv Rev. 2010. PMID: 20828588 Free PMC article. Review.
-
Emerging topics and new perspectives on HLA-G.Cell Mol Life Sci. 2011 Feb;68(3):433-51. doi: 10.1007/s00018-010-0584-3. Epub 2010 Nov 16. Cell Mol Life Sci. 2011. PMID: 21080027 Free PMC article. Review.
-
Inhibition of hepatic stellate cell proliferation by bone marrow mesenchymal stem cells via regulation of the cell cycle in rat.Exp Ther Med. 2012 Sep;4(3):375-380. doi: 10.3892/etm.2012.628. Epub 2012 Jun 29. Exp Ther Med. 2012. PMID: 23181102 Free PMC article.
-
From hepatocytes to stem and progenitor cells for liver regenerative medicine: advances and clinical perspectives.Cell Prolif. 2011 Apr;44 Suppl 1(Suppl 1):39-43. doi: 10.1111/j.1365-2184.2010.00730.x. Cell Prolif. 2011. PMID: 21481042 Free PMC article.
References
-
- Anjos‐Afonso F, Siapati EK, Bonnet D (2004) In vivo contribution of murine mesenchymal stem cells into multiple cell‐types under minimal damage conditions. J. Cell Sci. 117, 5655–5664. - PubMed
-
- Avital I, Inderbitzin D, Aoki T, Tyan DB, Cohen AH, Ferraresso C, Rozga J, Arnaout WS, Demetriou AA (2001) Isolation, characterization, and transplantation of bone marrow‐derived hepatocyte stem cells. Biochem. Biophys. Res. Commun. 288, 156–164. - PubMed
-
- Barry FP, Murphy JM (2004) Mesenchymal stem cells: clinical applications and biological characterization. Int. J. Biochem. Cell Biol. 36, 568–584. - PubMed
-
- Beerheide W, Von Mach MA, Ringel M, Fleckenstein C, Schumann S, Renzing N, Hildebrandt A, Brenner W, Jensen O, Gebhard S, Reifenberg K, Bender J, Oesch F, Hengstler JG (2002) Downregulation of beta2‐microglobulin in human cord blood somatic stem cells after transplantation into livers of SCID mice: an escape mechanism of stem cells? Biochem. Biophys. Res. Commun. 294, 1052–1063. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

