NPXY motifs in the beta1 integrin cytoplasmic tail are required for functional reovirus entry
- PMID: 18216114
- PMCID: PMC2268482
- DOI: 10.1128/JVI.01612-07
NPXY motifs in the beta1 integrin cytoplasmic tail are required for functional reovirus entry
Abstract
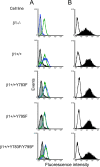
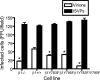
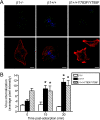
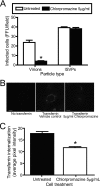
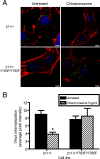
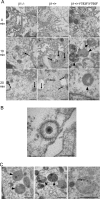
Reovirus cell entry is mediated by attachment to cell surface carbohydrate and junctional adhesion molecule A (JAM-A) and internalization by beta1 integrin. The beta1 integrin cytoplasmic tail contains two NPXY motifs, which function in recruitment of adaptor proteins and clathrin for endocytosis and serve as sorting signals for internalized cargo. As reovirus infection requires disassembly in the endocytic compartment, we investigated the role of the beta1 integrin NPXY motifs in reovirus internalization. In comparison to wild-type cells (beta1+/+ cells), reovirus infectivity was significantly reduced in cells expressing mutant beta1 integrin in which the NPXY motifs were altered to NPXF (beta1+/+Y783F/Y795F cells). However, reovirus displayed equivalent binding and internalization levels following adsorption to beta1+/+ cells and beta1+/+Y783F/Y795F cells, suggesting that the NPXY motifs are essential for transport of reovirus within the endocytic pathway. Reovirus entry into beta1+/+ cells was blocked by chlorpromazine, an inhibitor of clathrin-mediated endocytosis, while entry into beta1+/+Y783F/Y795F cells was unaffected. Furthermore, virus was distributed to morphologically distinct endocytic organelles in beta1+/+ and beta1+/+Y783F/Y795F cells, providing further evidence that the beta1 integrin NPXY motifs mediate sorting of reovirus in the endocytic pathway. Thus, NPXY motifs in the beta1 integrin cytoplasmic tail are required for functional reovirus entry, which indicates a key role for these sequences in endocytosis of a pathogenic virus.
Figures

Similar articles
-
Reovirus directly engages integrin to recruit clathrin for entry into host cells.Nat Commun. 2021 Apr 12;12(1):2149. doi: 10.1038/s41467-021-22380-0. Nat Commun. 2021. PMID: 33846319 Free PMC article.
-
Transport to late endosomes is required for efficient reovirus infection.J Virol. 2012 Aug;86(16):8346-58. doi: 10.1128/JVI.00100-12. Epub 2012 Jun 6. J Virol. 2012. PMID: 22674975 Free PMC article.
-
From touchdown to transcription: the reovirus cell entry pathway.Curr Top Microbiol Immunol. 2010;343:91-119. doi: 10.1007/82_2010_32. Curr Top Microbiol Immunol. 2010. PMID: 20397070 Free PMC article. Review.
-
Src kinase mediates productive endocytic sorting of reovirus during cell entry.J Virol. 2011 Apr;85(7):3203-13. doi: 10.1128/JVI.02056-10. Epub 2011 Jan 19. J Virol. 2011. PMID: 21248042 Free PMC article.
-
Reovirus receptors, cell entry, and proapoptotic signaling.Adv Exp Med Biol. 2013;790:42-71. doi: 10.1007/978-1-4614-7651-1_3. Adv Exp Med Biol. 2013. PMID: 23884585 Free PMC article. Review.
Cited by
-
SNX17 affects T cell activation by regulating TCR and integrin recycling.J Immunol. 2015 May 1;194(9):4555-66. doi: 10.4049/jimmunol.1402734. Epub 2015 Mar 30. J Immunol. 2015. PMID: 25825439 Free PMC article.
-
Forces during cellular uptake of viruses and nanoparticles at the ventral side.Nat Commun. 2020 Jan 2;11(1):32. doi: 10.1038/s41467-019-13877-w. Nat Commun. 2020. PMID: 31896744 Free PMC article.
-
Small peptide recognition sequence for intracellular sorting.Curr Opin Biotechnol. 2010 Oct;21(5):611-20. doi: 10.1016/j.copbio.2010.08.007. Curr Opin Biotechnol. 2010. PMID: 20817434 Free PMC article. Review.
-
ITGB1b-Deficient Rare Minnows Delay Grass Carp Reovirus (GCRV) Entry and Attenuate GCRV-Triggered Apoptosis.Int J Mol Sci. 2018 Oct 15;19(10):3175. doi: 10.3390/ijms19103175. Int J Mol Sci. 2018. PMID: 30326628 Free PMC article.
-
A modified lysosomal organelle mediates nonlytic egress of reovirus.J Cell Biol. 2020 Jul 6;219(7):e201910131. doi: 10.1083/jcb.201910131. J Cell Biol. 2020. PMID: 32356864 Free PMC article.
References
-
- Bansal, A., and L. M. Gierasch. 1991. The NPXY internalization signal of the LDL receptor adopts a reverse-turn conformation. Cell 671195-1201. - PubMed
-
- Barton, E. S., J. L. Connolly, J. C. Forrest, J. D. Chappell, and T. S. Dermody. 2001. Utilization of sialic acid as a coreceptor enhances reovirus attachment by multistep adhesion strengthening. J. Biol. Chem. 2762200-2211. - PubMed
-
- Barton, E. S., J. C. Forrest, J. L. Connolly, J. D. Chappell, Y. Liu, F. Schnell, A. Nusrat, C. A. Parkos, and T. S. Dermody. 2001. Junction adhesion molecule is a receptor for reovirus. Cell 104441-451. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

