Capture and enrichment of CD34-positive haematopoietic stem and progenitor cells from blood circulation using P-selectin in an implantable device
- PMID: 18218048
- PMCID: PMC2268974
- DOI: 10.1111/j.1365-2141.2007.06967.x
Capture and enrichment of CD34-positive haematopoietic stem and progenitor cells from blood circulation using P-selectin in an implantable device
Abstract
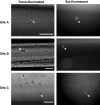
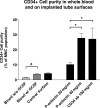
Clinical infusion of haematopoietic stem and progenitor cells (HSPCs) is vital for restoration of haematopoietic function in many cancer patients. Previously, we have demonstrated an ability to mimic physiological cell trafficking in order to capture CD34-positive (CD34+) HSPCs using monolayers of the cell adhesion protein P-selectin in flow chambers. The current study aimed to determine if HSPCs could be captured directly from circulating blood in vivo. Vascular shunt prototypes, coated internally with P-selectin, were inserted into the femoral artery of rats. Blood flow through the cell capture device resulted in a wall shear stress of 4-6 dynes/cm(2). After 1-h blood perfusion, immunofluorescence microscopy and flow cytometric analysis revealed successful capture of mononuclear cells positive for the HSPC surface marker CD34. Purity of captured CD34+ cells showed sevenfold enrichment over levels found in whole blood, with an average purity of 28%. Robust cell capture and HSPC enrichment were also demonstrated in devices that were implanted in a closed-loop arterio-venous shunt conformation for 2 h. Adherent cells were viable in culture and able to differentiate into burst-forming units. This study demonstrated an ability to mimic the physiological arrest of HSPCs from blood in an implantable device and may represent a practical alternative for adult stem cell capture and enrichment.
Figures


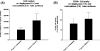

Similar articles
-
P-Selectin coated microtube for enrichment of CD34+ hematopoietic stem and progenitor cells from human bone marrow.Clin Chem. 2008 Jan;54(1):77-85. doi: 10.1373/clinchem.2007.089896. Epub 2007 Nov 16. Clin Chem. 2008. PMID: 18024531
-
Investigating the feasibility of stem cell enrichment mediated by immobilized selectins.Biotechnol Prog. 2007 Nov-Dec;23(6):1463-72. doi: 10.1021/bp0702222. Epub 2007 Oct 31. Biotechnol Prog. 2007. PMID: 17973400
-
Purification of CD45+ hematopoietic cells directly from human bone marrow using a flow-based P-selectin-coated microtube.Am J Hematol. 2008 Aug;83(8):627-9. doi: 10.1002/ajh.21204. Am J Hematol. 2008. PMID: 18442063
-
Red blood cell depletion and enrichment of CD34+ hematopoietic progenitor cells from human umbilical cord blood using soybean agglutinin and CD34 immunoselection.Exp Hematol. 1994 Nov;22(12):1134-40. Exp Hematol. 1994. PMID: 7523166
-
Positive selection of autologous peripheral blood stem cells.Baillieres Best Pract Res Clin Haematol. 1999 Mar-Jun;12(1-2):71-86. doi: 10.1053/beha.1999.0008. Baillieres Best Pract Res Clin Haematol. 1999. PMID: 11000984 Review.
Cited by
-
Stem cell enrichment with selectin receptors: mimicking the pH environment of trauma.Sensors (Basel). 2013 Sep 17;13(9):12516-26. doi: 10.3390/s130912516. Sensors (Basel). 2013. PMID: 24048341 Free PMC article.
-
A cell rolling cytometer reveals the correlation between mesenchymal stem cell dynamic adhesion and differentiation state.Lab Chip. 2014 Jan 7;14(1):161-6. doi: 10.1039/c3lc50923k. Lab Chip. 2014. PMID: 24146063 Free PMC article.
-
Heterogeneity of Circulating Tumor Cells in Breast Cancer: Identifying Metastatic Seeds.Int J Mol Sci. 2020 Mar 2;21(5):1696. doi: 10.3390/ijms21051696. Int J Mol Sci. 2020. PMID: 32121639 Free PMC article. Review.
-
Separation of SSEA-4 and TRA-1-60 labelled undifferentiated human embryonic stem cells from a heterogeneous cell population using magnetic-activated cell sorting (MACS) and fluorescence-activated cell sorting (FACS).Stem Cell Rev Rep. 2009 Mar;5(1):72-80. doi: 10.1007/s12015-009-9054-4. Epub 2009 Jan 31. Stem Cell Rev Rep. 2009. PMID: 19184635
-
P-selectin glycoprotein ligand-1 deficiency augments G-CSF induced myeloid cell mobilization.Naunyn Schmiedebergs Arch Pharmacol. 2014 Feb;387(2):109-18. doi: 10.1007/s00210-013-0913-9. Epub 2013 Oct 5. Naunyn Schmiedebergs Arch Pharmacol. 2014. PMID: 24091681
References
-
- Blair ML, Mickelsen D. Activation of lateral parabrachial nucleus neurons restores blood pressure and sympathetic vasomotor drive after hypotensive hemorrhage. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 2006a;291:R742–R750. - PubMed
-
- Blair ML, Mickelsen D. Plasma protein and blood volume restitution after hemorrhage in conscious pregnant and ovarian steroid-replaced rats. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology. 2006b;290:R425–R434. - PubMed
-
- Charles N, Liesveld JL, King MR. Investigating the feasibility of stem cell enrichment mediated by immobilized selectins. Biotechnology Progress. 2007;23:1463–1472. - PubMed
-
- Engelhardt M, Lubbert M, Guo Y. CD34(+) or CD34(−): which is the more primitive? Leukemia. 2002;16:1603–1608. - PubMed
-
- Fietz T, Rieger K, Dimeo F, Blau IW, Thiel E, Knauf WU. Stem cell mobilization in multiple myeloma patients: do we need an age-adjusted regimen for the elderly? Journal of Clinical Apheresis. 2004;19:202–207. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

