Tuberin haploinsufficiency is associated with the loss of OGG1 in rat kidney tumors
- PMID: 18218111
- PMCID: PMC2265742
- DOI: 10.1186/1476-4598-7-10
Tuberin haploinsufficiency is associated with the loss of OGG1 in rat kidney tumors
Abstract
Background: Tuberous sclerosis complex (TSC) is caused by defects in one of two tumor suppressor genes, TSC-1 or TSC-2. TSC-2 gene encodes tuberin, a protein involved in the pathogenesis of kidney tumors. Loss of heterozygosity (LOH) at the TSC2 locus has been detected in TSC-associated renal cell carcinoma (RCC) and in RCC in the Eker rat. Tuberin downregulates the DNA repair enzyme 8-oxoguanine DNA-glycosylase (OGG1) with important functional consequences, compromising the ability of cells to repair damaged DNA resulting in the accumulation of the mutagenic oxidized DNA, 8-oxo-dG. Loss of function mutations of OGG1 also occurs in human kidney clear cell carcinoma and may contribute to tumorgenesis. We investigated the distribution of protein expression and the activity of OGG1 and 8-oxo-dG and correlated it with the expression of tuberin in kidneys of wild type and Eker rats and tumor from Eker rat.
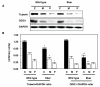
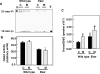
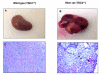
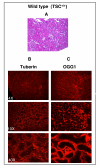
Results: Tuberin expression, OGG1 protein expression and activity were higher in kidney cortex than in medulla or papilla in both wild type and Eker rats. On the other hand, 8-oxo-dG levels were highest in the medulla, which expressed the lowest levels of OGG1. The basal levels of 8-oxo-dG were also higher in both cortex and medulla of Eker rats compared to wild type rats. In kidney tumors from Eker rats, the loss of the second TSC2 allele is associated with loss of OGG1 expression. Immunostaining of kidney tissue shows localization of tuberin and OGG1 mainly in the cortex.
Conclusion: These results demonstrate that OGG1 localizes with tuberin preferentially in kidney cortex. Loss of tuberin is accompanied by the loss of OGG1 contributing to tumorgenesis. In addition, the predominant expression of OGG1 in the cortex and its decreased expression and activity in the Eker rat may account for the predominant cortical localization of renal cell carcinoma.
Figures


Similar articles
-
Insight into mechanism of oxidative DNA damage in angiomyolipomas from TSC patients.Mol Cancer. 2009 Mar 5;8:13. doi: 10.1186/1476-4598-8-13. Mol Cancer. 2009. PMID: 19265534 Free PMC article.
-
Novel mechanism of regulation of the DNA repair enzyme OGG1 in tuberin-deficient cells.Carcinogenesis. 2010 Nov;31(11):2022-30. doi: 10.1093/carcin/bgq189. Epub 2010 Sep 13. Carcinogenesis. 2010. PMID: 20837600 Free PMC article.
-
Reduced constitutive 8-oxoguanine-DNA glycosylase expression and impaired induction following oxidative DNA damage in the tuberin deficient Eker rat.Carcinogenesis. 2003 Mar;24(3):573-82. doi: 10.1093/carcin/24.3.573. Carcinogenesis. 2003. PMID: 12663520
-
Tuberous sclerosis complex and DNA repair.Adv Exp Med Biol. 2010;685:84-94. doi: 10.1007/978-1-4419-6448-9_8. Adv Exp Med Biol. 2010. PMID: 20687497 Review.
-
Tuberous sclerosis as an underlying basis for infantile spasm.Int Rev Neurobiol. 2002;49:315-32. doi: 10.1016/s0074-7742(02)49019-8. Int Rev Neurobiol. 2002. PMID: 12040899 Review.
Cited by
-
Genetics and molecular biology of tuberous sclerosis complex.Curr Genomics. 2008 Nov;9(7):475-87. doi: 10.2174/138920208786241243. Curr Genomics. 2008. PMID: 19506736 Free PMC article.
-
Tuberin inhibits production of the matrix protein fibronectin in diabetes.J Am Soc Nephrol. 2012 Oct;23(10):1652-62. doi: 10.1681/ASN.2012030285. Epub 2012 Aug 16. J Am Soc Nephrol. 2012. PMID: 22904348 Free PMC article.
-
Insight into mechanism of oxidative DNA damage in angiomyolipomas from TSC patients.Mol Cancer. 2009 Mar 5;8:13. doi: 10.1186/1476-4598-8-13. Mol Cancer. 2009. PMID: 19265534 Free PMC article.
-
The tuberin/mTOR pathway promotes apoptosis of tubular epithelial cells in diabetes.J Am Soc Nephrol. 2011 Feb;22(2):262-73. doi: 10.1681/ASN.2010040352. J Am Soc Nephrol. 2011. PMID: 21289215 Free PMC article.
-
Novel mechanism of regulation of the DNA repair enzyme OGG1 in tuberin-deficient cells.Carcinogenesis. 2010 Nov;31(11):2022-30. doi: 10.1093/carcin/bgq189. Epub 2010 Sep 13. Carcinogenesis. 2010. PMID: 20837600 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

