Inhibition of apoptosis in neuronal cells infected with Chlamydophila (Chlamydia) pneumoniae
- PMID: 18218130
- PMCID: PMC2266938
- DOI: 10.1186/1471-2202-9-13
Inhibition of apoptosis in neuronal cells infected with Chlamydophila (Chlamydia) pneumoniae
Abstract
Background: Chlamydophila (Chlamydia) pneumoniae is an intracellular bacterium that has been identified within cells in areas of neuropathology found in Alzheimer disease (AD), including endothelia, glia, and neurons. Depending on the cell type of the host, infection by C. pneumoniae has been shown to influence apoptotic pathways in both pro- and anti-apoptotic fashions. We have hypothesized that persistent chlamydial infection of neurons may be an important mediator of the characteristic neuropathology observed in AD brains. Chronic and/or persistent infection of neuronal cells with C. pneumoniae in the AD brain may affect apoptosis in cells containing chlamydial inclusions.
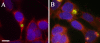
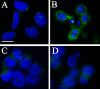
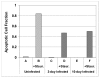
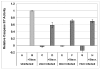
Results: SK-N-MC neuroblastoma cells were infected with the respiratory strain of C. pneumoniae, AR39 at an MOI of 1. Following infection, the cells were either untreated or treated with staurosporine and then examined for apoptosis by labeling for nuclear fragmentation, caspase activity, and membrane inversion as indicated by annexin V staining. C. pneumoniae infection was maintained through 10 days post-infection. At 3 and 10 days post-infection, the infected cell cultures appeared to inhibit or were resistant to the apoptotic process when induced by staurosporine. This inhibition was demonstrated quantitatively by nuclear profile counts and caspase 3/7 activity measurements.
Conclusion: These data suggest that C. pneumoniae can sustain a chronic infection in neuronal cells by interfering with apoptosis, which may contribute to chronic inflammation in the AD brain.
Figures




Similar articles
-
Epithelial cells infected with Chlamydophila pneumoniae (Chlamydia pneumoniae) are resistant to apoptosis.Infect Immun. 2001 Dec;69(12):7880-8. doi: 10.1128/IAI.69.12.7880-7888.2001. Infect Immun. 2001. PMID: 11705971 Free PMC article.
-
Chlamydia pneumoniae induces aponecrosis in human aortic smooth muscle cells.BMC Microbiol. 2005 Jan 21;5:2. doi: 10.1186/1471-2180-5-2. BMC Microbiol. 2005. PMID: 15663783 Free PMC article.
-
Chlamydia pneumoniae inhibits apoptosis in human epithelial and monocyte cell lines.Scand J Immunol. 2002 Apr;55(4):390-8. doi: 10.1046/j.1365-3083.2002.01075.x. Scand J Immunol. 2002. PMID: 11967121
-
Chlamydia pneumoniae infection and Alzheimer's disease: a connection to remember?Med Microbiol Immunol. 2010 Nov;199(4):283-9. doi: 10.1007/s00430-010-0162-1. Epub 2010 May 6. Med Microbiol Immunol. 2010. PMID: 20445987 Review.
-
Characterization of antiapoptotic activities of Chlamydia pneumoniae in infected cells.Ann N Y Acad Sci. 2003 Dec;1010:565-7. doi: 10.1196/annals.1299.105. Ann N Y Acad Sci. 2003. PMID: 15033792 Review.
Cited by
-
Chronic Lyme Disease and Co-infections: Differential Diagnosis.Open Neurol J. 2012;6:158-78. doi: 10.2174/1874205X01206010158. Epub 2012 Dec 28. Open Neurol J. 2012. PMID: 23400696 Free PMC article.
-
The Possibility of an Infectious Etiology of Alzheimer Disease.Mol Neurobiol. 2019 Jun;56(6):4479-4491. doi: 10.1007/s12035-018-1388-y. Epub 2018 Oct 18. Mol Neurobiol. 2019. PMID: 30338482 Review.
-
Herpes Simplex Virus Type 1 and Other Pathogens are Key Causative Factors in Sporadic Alzheimer's Disease.J Alzheimers Dis. 2015;48(2):319-53. doi: 10.3233/JAD-142853. J Alzheimers Dis. 2015. PMID: 26401998 Free PMC article. Review.
-
The Role of Genetic, Environmental, and Dietary Factors in Alzheimer's Disease: A Narrative Review.Int J Mol Sci. 2025 Jan 30;26(3):1222. doi: 10.3390/ijms26031222. Int J Mol Sci. 2025. PMID: 39940989 Free PMC article. Review.
-
Chlamydia pneumoniae in Alzheimer's disease pathology.Front Neurosci. 2024 May 6;18:1393293. doi: 10.3389/fnins.2024.1393293. eCollection 2024. Front Neurosci. 2024. PMID: 38770241 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

