Anti-cancer effects of JKA97 are associated with its induction of cell apoptosis via a Bax-dependent and p53-independent pathway
- PMID: 18218619
- PMCID: PMC2417161
- DOI: 10.1074/jbc.M707860200
Anti-cancer effects of JKA97 are associated with its induction of cell apoptosis via a Bax-dependent and p53-independent pathway
Erratum in
- J Biol Chem. 2012 Jul 6;287(28):23850
Abstract
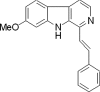
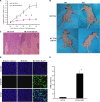
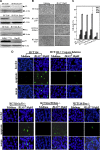
p53, one of the most commonly mutated genes in human cancers, is thought to be associated with cancer development. Hence, screening and identifying natural or synthetic compounds with anti-cancer activity via p53-independent pathway is one of the most challenging tasks for scientists in this field. Compound JKA97 (methoxy-1-styryl-9H-pyrid-[3,4-b]-indole) is a small molecule synthetic anti-cancer agent, with unknown mechanism(s). In this study we have demonstrated that the anti-cancer activity of JKA97 is associated with apoptotic induction via p53-independent mechanisms. We found that co-incubation of human colon cancer HCT116 cells with JKA97 inhibited HCT116 cell anchorage-independent growth in vitro and tumorigenicity in nude mice and also induced a cell apoptotic response, both in the cell culture model and in a tumorigenesis nude mouse model. Further studies showed that JKA97-induced apoptosis was dramatically impaired in Bax knock-out (Bax(-/-)) HCT116 cells, whereas the knock-out of p53 or PUMA did not show any inhibitory effects. The p53-independent apoptotic induction by JKA97 was confirmed in other colon cancer and hepatocarcinoma cell lines. In addition, our results showed an induction of Bax translocation and cytochrome c release from the mitochondria to the cytosol in HCT116 cells, demonstrating that the compound induces apoptosis through a Bax-initiated mitochondria-dependent pathway. These studies provide a molecular basis for the therapeutic application of JKA97 against human cancers with p53 mutations.
Figures




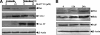
Similar articles
-
JKA97, a novel benzylidene analog of harmine, exerts anti-cancer effects by inducing G1 arrest, apoptosis, and p53-independent up-regulation of p21.PLoS One. 2012;7(4):e34303. doi: 10.1371/journal.pone.0034303. Epub 2012 Apr 27. PLoS One. 2012. PMID: 22558087 Free PMC article.
-
Hsp90 inhibitors promote p53-dependent apoptosis through PUMA and Bax.Mol Cancer Ther. 2013 Nov;12(11):2559-68. doi: 10.1158/1535-7163.MCT-13-0284. Epub 2013 Aug 21. Mol Cancer Ther. 2013. PMID: 23966620 Free PMC article.
-
6-Hydroxydopamine activates the mitochondrial apoptosis pathway through p38 MAPK-mediated, p53-independent activation of Bax and PUMA.J Neurochem. 2008 Mar;104(6):1599-612. doi: 10.1111/j.1471-4159.2007.05115.x. Epub 2007 Nov 6. J Neurochem. 2008. PMID: 17996028
-
Purified vitexin compound 1, a new neolignan isolated compound, promotes PUMA-dependent apoptosis in colorectal cancer.Cancer Med. 2018 Dec;7(12):6158-6169. doi: 10.1002/cam4.1769. Epub 2018 Nov 6. Cancer Med. 2018. PMID: 30402948 Free PMC article.
-
Exploring the dual role of SIVA1 in cancer biology.Gene. 2025 May 20;950:149365. doi: 10.1016/j.gene.2025.149365. Epub 2025 Feb 28. Gene. 2025. PMID: 40024298 Review.
Cited by
-
XIAP BIR domain suppresses miR-200a expression and subsequently promotes EGFR protein translation and anchorage-independent growth of bladder cancer cell.J Hematol Oncol. 2017 Jan 5;10(1):6. doi: 10.1186/s13045-016-0376-9. J Hematol Oncol. 2017. PMID: 28057023 Free PMC article.
-
SUMOylation of RhoGDIα is required for its repression of cyclin D1 expression and anchorage-independent growth of cancer cells.Mol Oncol. 2014 Mar;8(2):285-96. doi: 10.1016/j.molonc.2013.11.006. Epub 2013 Dec 3. Mol Oncol. 2014. PMID: 24342356 Free PMC article.
-
Isorhapontigenin (ISO) inhibited cell transformation by inducing G0/G1 phase arrest via increasing MKP-1 mRNA Stability.Oncotarget. 2014 May 15;5(9):2664-77. doi: 10.18632/oncotarget.1872. Oncotarget. 2014. PMID: 24797581 Free PMC article.
-
MicroRNA-411 Downregulation Enhances Tumor Growth by Upregulating MLLT11 Expression in Human Bladder Cancer.Mol Ther Nucleic Acids. 2018 Jun 1;11:312-322. doi: 10.1016/j.omtn.2018.03.003. Epub 2018 Mar 10. Mol Ther Nucleic Acids. 2018. PMID: 29858066 Free PMC article.
-
Thymoquinone Selectively Induces Hepatocellular Carcinoma Cell Apoptosis in Synergism With Clinical Therapeutics and Dependence of p53 Status.Front Pharmacol. 2020 Sep 15;11:555283. doi: 10.3389/fphar.2020.555283. eCollection 2020. Front Pharmacol. 2020. PMID: 33041795 Free PMC article.
References
-
- Svec, J. (2005) Bratisl. Lek. Listy 106 238-239 - PubMed
-
- Lane, D. P. (1993) Nature 362 786-787 - PubMed
-
- Wang, J., Ouyang, W., Li, J., Wei, L., Ma, Q., Zhang, Z., Tong, Q., He, J., and Huang, C. (2005) Cancer Res. 65 6601-6611 - PubMed
-
- Tweddle, D. A., Pearson, A. D., Haber, M., Norris, M. D., Xue, C., Flemming, C., and Lunec, J. (2003) Cancer Lett. 197 93-98 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

