Structure-function analyses involving palindromic analogs of tritrypticin suggest autonomy of anti-endotoxin and antibacterial activities
- PMID: 18218719
- PMCID: PMC2248310
- DOI: 10.1110/ps.073145008
Structure-function analyses involving palindromic analogs of tritrypticin suggest autonomy of anti-endotoxin and antibacterial activities
Abstract
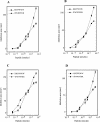
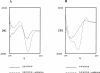
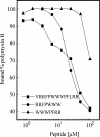
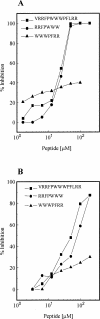
Neutralization of invading pathogens by gene-encoded peptide antibiotics has been suggested to manifest in a variety of different modes. Some of these modes require internalization of the peptide through a pathway that involves LPS-mediated uptake of the peptide antibiotics. Many proline/tryptophan-rich cationic peptides for which this mode has been invoked do, indeed, show LPS (endotoxin) binding. If the mechanism of antibiotic action involves the LPS-mediated pathway, a positive correlation ought to manifest between the binding to LPS, its neutralization, and the bacterial killing. No such correlation was evident based on our studies involving minimal active analogs of tritrypticin. The anti-endotoxin activities of these analogs appear not to relate directly to their antibiotic potential. The two palindromic analogs of tritrypticin, NT7 (RRFPWWW) and CT7 (WWWPFRR), showed comparable antibacterial activities. However, while NT7 exhibited anti-endotoxin activity, CT7 did not. The LPS binding of two tritrypticin analogs correlated with their corresponding structures, but the antibacterial activities did not. Further structure-function analysis indicated specific structural implications of the antibacterial activity at the molecular level. Studies involving designed analogs of NT7 incorporating either rigid or flexible linkers between the specifically distanced hydrophobic and cationic clusters modulate the LPS binding. On the other hand, not knowing the target receptor for antibacterial activity is a drawback since the precise epitope for antibacterial activity is not definable. It is apparent that the anti-endotoxin and antibacterial activities represent two independent functions of tritrypticin, consistent with the emerging multifunctionality in the nature of cathelicidins.
Figures

Similar articles
-
Structure-function analysis of tritrypticin, an antibacterial peptide of innate immune origin.J Biol Chem. 1999 Aug 13;274(33):23296-304. doi: 10.1074/jbc.274.33.23296. J Biol Chem. 1999. PMID: 10438505
-
Endotoxin-binding synthetic peptides with endotoxin-neutralizing, antibacterial and anticoagulant activities.Prog Clin Biol Res. 1994;388:147-59. Prog Clin Biol Res. 1994. PMID: 7831355
-
Plasticity in structure and interactions is critical for the action of indolicidin, an antibacterial peptide of innate immune origin.Protein Sci. 2002 Sep;11(9):2158-67. doi: 10.1110/ps.0211602. Protein Sci. 2002. PMID: 12192071 Free PMC article.
-
De novo designed lipopolysaccharide binding peptides: structure based development of antiendotoxic and antimicrobial drugs.Curr Med Chem. 2010;17(27):3080-93. doi: 10.2174/092986710791959756. Curr Med Chem. 2010. PMID: 20629624 Review.
-
Peptides with dual mode of action: Killing bacteria and preventing endotoxin-induced sepsis.Biochim Biophys Acta. 2016 May;1858(5):971-9. doi: 10.1016/j.bbamem.2016.01.011. Epub 2016 Jan 20. Biochim Biophys Acta. 2016. PMID: 26801369 Review.
Cited by
-
Putative bioactive motif of tritrpticin revealed by an antibody with biological receptor-like properties.PLoS One. 2013 Sep 24;8(9):e75582. doi: 10.1371/journal.pone.0075582. eCollection 2013. PLoS One. 2013. PMID: 24086578 Free PMC article.
-
In Vivo, In Vitro, and In Silico Characterization of Peptoids as Antimicrobial Agents.PLoS One. 2016 Feb 5;11(2):e0135961. doi: 10.1371/journal.pone.0135961. eCollection 2016. PLoS One. 2016. PMID: 26849681 Free PMC article.
References
-
- Casteels, P., Tempst, P. Apidaecin-type peptide antibiotics function through a non-poreforming mechanism involving stereospecificity. Biochem. Biophys. Res. Commun. 1994;199:339–345. - PubMed
-
- de Haas, C.J., Haas, P.J., van Kessel, K.P., van Strijp, J.A. Affinities of different proteins and peptides for lipopolysaccharide as determined by biosensor technology. Biochem. Biophys. Res. Commun. 1998;252:492–496. - PubMed
-
- Falla, T.J., Karunaratne, D.N., Hancock, R.E.W. Mode of action of the antimicrobial peptide Indolicidin. J. Biol. Chem. 1996;271:19298–19303. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
