MR thermometry
- PMID: 18219673
- PMCID: PMC2780364
- DOI: 10.1002/jmri.21265
MR thermometry
Abstract
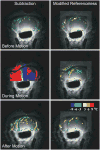
Minimally invasive thermal therapy as local treatment of benign and malignant diseases has received increasing interest in recent years. Safety and efficacy of the treatment require accurate temperature measurement throughout the thermal procedure. Noninvasive temperature monitoring is feasible with magnetic resonance (MR) imaging based on temperature-sensitive MR parameters such as the proton resonance frequency (PRF), the diffusion coefficient (D), T1 and T2 relaxation times, magnetization transfer, the proton density, as well as temperature-sensitive contrast agents. In this article the principles of temperature measurements with these methods are reviewed and their usefulness for monitoring in vivo procedures is discussed. Whereas most measurements give a temperature change relative to a baseline condition, temperature-sensitive contrast agents and spectroscopic imaging can provide absolute temperature measurements. The excellent linearity and temperature dependence of the PRF and its near independence of tissue type have made PRF-based phase mapping methods the preferred choice for many in vivo applications. Accelerated MRI imaging techniques for real-time monitoring with the PRF method are discussed. Special attention is paid to acquisition and reconstruction methods for reducing temperature measurement artifacts introduced by tissue motion, which is often unavoidable during in vivo applications.
(Copyright) 2008 Wiley-Liss, Inc.
Figures



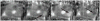
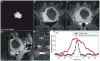
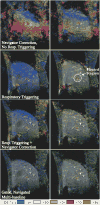
References
-
- Kim JH, Hahn EW. Clinical and biological studies of localized hyperthermia. Cancer Res. 1979;39:2258–2261. - PubMed
-
- Thomsen S. Pathologic analysis of photothermal and photomechanical effects of laser-tissue interactions. Photochem Photobiol. 1991;53:825–835. - PubMed
-
- Sapareto SA, Dewey WC. Thermal dose determination in cancer therapy. Int J Radiat Oncol Biol Phys. 1984;10:787–800. - PubMed
-
- Dewhirst MW, Viglianti BL, Lora-Michiels M, Hanson M, Hoopes PJ. Basic principles of thermal dosimetry and thermal thresholds for tissue damage from hyperthermia. Int J Hyperthermia. 2003;19:267–294. - PubMed
-
- Mack MG, Straub R, Eichler K, Engelmann K, Zangos S, Roggan A, Woitaschek D, Bottger M, Vogl TJ. Percutaneous MR imaging-guided laser-induced thermotherapy of hepatic metastases. Abdom Imaging. 2001;26:369–374. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

