Vesicle-independent extracellular release of a proinflammatory outer membrane lipoprotein in free-soluble form
- PMID: 18226201
- PMCID: PMC2257964
- DOI: 10.1186/1471-2180-8-18
Vesicle-independent extracellular release of a proinflammatory outer membrane lipoprotein in free-soluble form
Abstract
Background: Aggregatibacter actinomycetemcomitans is an oral bacterium associated with aggressively progressing periodontitis. Extracellular release of bacterial outer membrane proteins has been suggested to mainly occur via outer membrane vesicles. This study investigated the presence and conservation of peptidoglycan-associated lipoprotein (AaPAL) among A. actinomycetemcomitans strains, the immunostimulatory effect of AaPAL, and whether live cells release this structural outer membrane lipoprotein in free-soluble form independent of vesicles.
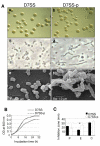
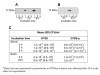
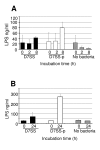
Results: The pal locus and its gene product were confirmed in clinical A. actinomycetemcomitans strains by PCR-restriction fragment length polymorphism and immunoblotting. Culturing under different growth conditions revealed no apparent requirement for the AaPAL expression. Inactivation of pal in a wild-type strain (D7S) and in its spontaneous laboratory variant (D7SS) resulted in pleiotropic cellular effects. In a cell culture insert model (filter pore size 0.02 mum), AaPAL was detected from filtrates when strains D7S and D7SS were incubated in serum or broth in the inserts. Electron microscopy showed that A. actinomycetemcomitans vesicles (0.05-0.2 mum) were larger than the filter pores and that there were no vesicles in the filtrates. The filtrates were immunoblot negative for a cytoplasmic marker, cyclic AMP (cAMP) receptor protein. An ex vivo model indicated cytokine production from human whole blood stimulated by AaPAL.
Conclusion: Free-soluble AaPAL can be extracellularly released in a process independent of vesicles.
Figures






References
-
- Norskov-Lauritsen N, Kilian M. Reclassification of Actinobacillus actinomycetemcomitans, Haemophilus aphrophilus, Haemophilus paraphrophilus and Haemophilus segnis as Aggregatibacter actinomycetemcomitans gen. nov., comb. nov., Aggregatibacter aphrophilus comb. nov. and Aggregatibacter segnis comb. nov., and emended description of Aggregatibacter aphrophilus to include V factor-dependent and V factor-independent isolates . Int J Syst Evol Microbiol. 2006;56:2135–2146. doi: 10.1099/ijs.0.64207-0. - DOI - PubMed
-
- Socransky SS, Haffajee AD. The bacterial etiology of destructive periodontal disease: current concepts. J Periodontol. 1992;63:322–331. - PubMed
-
- Paul-Satyaseela M, Karched M, Bian Z, Ihalin R, Boren T, Arnqvist A, Chen C, Asikainen S. Immunoproteomics of Actinobacillus actinomycetemcomitans outer-membrane proteins reveal a highly immunoreactive peptidoglycan-associated lipoprotein . J Med Microbiol. 2006;55:931–942. doi: 10.1099/jmm.0.46470-0. - DOI - PubMed
-
- Sturgis JN. Organisation and evolution of the tol-pal gene cluster. J Mol Microbiol Biotechnol. 2001;3:113–122. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

