Sublingual vaccination with influenza virus protects mice against lethal viral infection
- PMID: 18227512
- PMCID: PMC2234198
- DOI: 10.1073/pnas.0708684105
Sublingual vaccination with influenza virus protects mice against lethal viral infection
Abstract
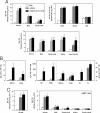
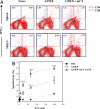
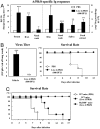
We assessed whether the sublingual (s.l.) route would be an effective means of delivering vaccines against influenza virus in mice by using either formalin-inactivated or live influenza A/PR/8 virus (H1N1). Sublingual administration of inactivated influenza virus given on two occasions induced both systemic and mucosal antibody responses and conferred protection against a lethal intranasal (i.n.) challenge with influenza virus. Coadministration of a mucosal adjuvant (mCTA-LTB) enhanced these responses and resulted in complete protection against respiratory viral challenge. In addition, s.l. administration of formalin-inactivated A/PR/8 plus mCTA-LTB induced systemic expansion of IFN-gamma-secreting T cells and virus-specific cytotoxic T lymphocyte responses. Importantly, a single s.l. administration of live A/PR/8 virus was not pathogenic and induced protection mediated by both acquired and innate immunity. Moreover, s.l. administration of live A/PR/8 virus conferred heterosubtypic protection against respiratory challenge with H3N2 virus. Unlike the i.n. route, the A/PR/8 virus, whether live or inactivated, did not migrate to or replicate in the CNS after s.l. administration. Based on these promising findings, we propose that the s.l. mucosal route offers an attractive alternative to mucosal routes for administering influenza vaccines.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Heterosubtypic cross-protection induced by whole inactivated influenza virus vaccine in mice: influence of the route of vaccine administration.Influenza Other Respir Viruses. 2013 Nov;7(6):1202-9. doi: 10.1111/irv.12142. Epub 2013 Sep 16. Influenza Other Respir Viruses. 2013. PMID: 24102979 Free PMC article.
-
Induction of heterosubtypic immunity to influenza virus by intranasal immunization.J Virol. 2008 Feb;82(3):1350-9. doi: 10.1128/JVI.01615-07. Epub 2007 Nov 21. J Virol. 2008. PMID: 18032492 Free PMC article.
-
Bacterium-like particles supplemented with inactivated influenza antigen induce cross-protective influenza-specific antibody responses through intranasal administration.Vaccine. 2012 Jul 6;30(32):4884-91. doi: 10.1016/j.vaccine.2012.04.032. Epub 2012 Apr 23. Vaccine. 2012. PMID: 22537989
-
Intranasal immunization with influenza VLPs incorporating membrane-anchored flagellin induces strong heterosubtypic protection.PLoS One. 2010 Nov 29;5(11):e13972. doi: 10.1371/journal.pone.0013972. PLoS One. 2010. PMID: 21124769 Free PMC article.
-
Stressor-induced alterations of adaptive immunity to vaccination and viral pathogens.Immunol Allergy Clin North Am. 2011 Feb;31(1):69-79. doi: 10.1016/j.iac.2010.09.002. Immunol Allergy Clin North Am. 2011. PMID: 21094924 Free PMC article. Review.
Cited by
-
Microneedle-Mediated Vaccine Delivery to the Oral Mucosa.Adv Healthc Mater. 2019 Feb;8(4):e1801180. doi: 10.1002/adhm.201801180. Epub 2018 Dec 10. Adv Healthc Mater. 2019. PMID: 30537400 Free PMC article. Review.
-
Inactivated Eyedrop Influenza Vaccine Adjuvanted with Poly(I:C) Is Safe and Effective for Inducing Protective Systemic and Mucosal Immunity.PLoS One. 2015 Sep 10;10(9):e0137608. doi: 10.1371/journal.pone.0137608. eCollection 2015. PLoS One. 2015. PMID: 26355295 Free PMC article.
-
Update on Chlamydia trachomatis Vaccinology.Clin Vaccine Immunol. 2017 Apr 5;24(4):e00543-16. doi: 10.1128/CVI.00543-16. Print 2017 Apr. Clin Vaccine Immunol. 2017. PMID: 28228394 Free PMC article. Review.
-
Tuning Helical Peptide Nanofibers as a Sublingual Vaccine Platform for a Variety of Peptide Epitopes.Adv Healthc Mater. 2025 Jan;14(3):e2402055. doi: 10.1002/adhm.202402055. Epub 2024 Dec 15. Adv Healthc Mater. 2025. PMID: 39676346
-
Robust antigen-specific humoral immune responses to sublingually delivered adenoviral vectors encoding HIV-1 Env: association with mucoadhesion and efficient penetration of the sublingual barrier.Vaccine. 2011 Sep 16;29(40):7080-9. doi: 10.1016/j.vaccine.2011.07.008. Epub 2011 Jul 27. Vaccine. 2011. PMID: 21801777 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

