Influenza: the virus and prophylaxis with inactivated influenza vaccine in "at risk" groups, including COPD patients
- PMID: 18229561
- PMCID: PMC2695195
Influenza: the virus and prophylaxis with inactivated influenza vaccine in "at risk" groups, including COPD patients
Abstract
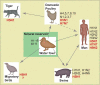
Influenza is a major respiratory pathogen, which exerts a huge human and economic toll on society. Influenza is a vaccine preventable disease, however, the vaccine strains must be annually updated due to the continuous antigenic changes in the virus. Inactivated influenza vaccines have been used for over 50 years and have an excellent safety record. Annual vaccination is therefore recommended for all individuals with serious medical conditions, like COPD, and protects the vaccinee against influenza illness and also against hospitalization and death. In COPD patients, influenza infection can lead to exacerbations resulting in reduced quality of life, hospitalization and death in the most severe cases. Although there is only limited literature on the use of influenza vaccination solely in COPD patients, there is clearly enough evidence to recommend annual vaccination in this group. This review will focus on influenza virus and prophylaxis with inactivated influenza vaccines in COPD patients and other "at risk" groups to reduce morbidity, save lives, and reduce health care costs.
Figures

References
-
- Barry DW, Mayner RE, Staton E, et al. Comparative trial of influenza vaccines. I. Immunogenicity of whole virus and split product vaccines in man. Am J Epidemiol. 1976;104:34–46. - PubMed
-
- Baydur A. Influenza vaccination in vulnerable populations. Chest. 2004;125:1971–2. - PubMed
-
- Beyer WE, de Bruijn IA, Palache AM, et al. Protection against influenza after annually repeated vaccination: a meta-analysis of serologic and field studies. Arch Intern Med. 1999;159:182–8. - PubMed
-
- Beyer WE, Palache AM, de Jong JC, et al. Cold-adapted live influenza vaccine versus inactivated vaccine: systemic vaccine reactions, local and systemic antibody response, and vaccine efficacy. A meta-analysis. Vaccine. 2002;20:1340–53. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

