Inhibitory Effects of Conjugated Epicatechin Metabolites on Peroxynitrite-mediated Nitrotyrosine Formation
- PMID: 18231630
- PMCID: PMC2212348
- DOI: 10.3164/jcbn.2008008
Inhibitory Effects of Conjugated Epicatechin Metabolites on Peroxynitrite-mediated Nitrotyrosine Formation
Abstract
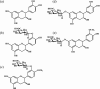
Previously, we identified four metabolites of (-)-epicatechin in blood and urine: (-)-epicatechin-3'-O-glucuronide (E3'G), 4'-O-methyl-(-)-epicatechin-3'-O-glucuronide (4'ME3'G), (-)-epicatechin-7-O-glucuronide (E7G), and 3'-O-methyl-(-)-epicatechin-7-O-glucuronide (3'ME7G) (Natsume et al. Free Radical Biol. Med. 34, 840-849, 2003). The aim of the current study was to compare the antioxidative activities of these metabolites with that of their parent compound. After oral administration of (-)-epicatechin, E3'G and 4'ME3'G were isolated from human urine, and E7G and 3'ME7G isolated from rat urine. We found that these compounds inhibited peroxynitrite-mediated tyrosine nitration, in the following order of potency: E3'G > (-)-epicatechin > E7G = 3'ME7G. = 4'ME3'G. These results demonstrate that the metabolites of (-)-epicatechin retain antioxidative activity on peroxynitrite-induced oxidative damages to some extent.
Keywords: (−)-epicatechin; glucuronide; metabolites; nitrotyrosine; peroxynitrite.
Figures

Similar articles
-
In vitro antioxidative activity of (-)-epicatechin glucuronide metabolites present in human and rat plasma.Free Radic Res. 2004 Dec;38(12):1341-8. doi: 10.1080/10715760400022087. Free Radic Res. 2004. PMID: 15763958
-
Structures of (-)-epicatechin glucuronide identified from plasma and urine after oral ingestion of (-)-epicatechin: differences between human and rat.Free Radic Biol Med. 2003 Apr 1;34(7):840-9. doi: 10.1016/s0891-5849(02)01434-x. Free Radic Biol Med. 2003. PMID: 12654472
-
Identification of (-)-epicatechin metabolites and their metabolic fate in the rat.Drug Metab Dispos. 1999 Feb;27(2):309-16. Drug Metab Dispos. 1999. PMID: 9929521
-
Absorption, metabolism, distribution and excretion of (-)-epicatechin: A review of recent findings.Mol Aspects Med. 2018 Jun;61:18-30. doi: 10.1016/j.mam.2017.11.002. Epub 2017 Nov 9. Mol Aspects Med. 2018. PMID: 29126853 Review.
-
Peroxynitrite-dependent modifications of tyrosine residues in hemoglobin. Formation of tyrosyl radical(s) and 3-nitrotyrosine.Amino Acids. 2003 Dec;25(3-4):341-50. doi: 10.1007/s00726-003-0021-0. Epub 2003 Sep 4. Amino Acids. 2003. PMID: 14661095 Review.
Cited by
-
Metabolic conversion of dietary flavonoids alters their anti-inflammatory and antioxidant properties.Free Radic Biol Med. 2011 Jul 15;51(2):454-63. doi: 10.1016/j.freeradbiomed.2011.04.032. Epub 2011 Apr 22. Free Radic Biol Med. 2011. PMID: 21571063 Free PMC article.
-
Epigallocatechin Gallate: A Review of Its Beneficial Properties to Prevent Metabolic Syndrome.Nutrients. 2015 Jul 7;7(7):5443-68. doi: 10.3390/nu7075230. Nutrients. 2015. PMID: 26198245 Free PMC article. Review.
-
Chinmedomics Strategy for Elucidating the Pharmacological Effects and Discovering Bioactive Compounds From Keluoxin Against Diabetic Retinopathy.Front Pharmacol. 2022 Mar 31;13:728256. doi: 10.3389/fphar.2022.728256. eCollection 2022. Front Pharmacol. 2022. PMID: 35431942 Free PMC article.
-
Wine Flavonoids in Health and Disease Prevention.Molecules. 2017 Feb 14;22(2):292. doi: 10.3390/molecules22020292. Molecules. 2017. PMID: 28216567 Free PMC article. Review.
-
Polyphenols Beyond Barriers: A Glimpse into the Brain.Curr Neuropharmacol. 2017;15(4):562-594. doi: 10.2174/1570159X14666161026151545. Curr Neuropharmacol. 2017. PMID: 27784225 Free PMC article. Review.
References
-
- Scalbert A., Williamson G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000;130:2073S–2085S. - PubMed
-
- Natsume M., Osakabe N., Yamagishi M., Takizawa T., Nakamura T., Miyatake H., Hatano T., Yoshida T. Analyses of polyphenols in cacao liquor, cocoa, and chocolate by normal- phase and reversed-phase HPLC. Biosci. Biotechnol. Biochem. 2000;64:2581–2587. - PubMed
-
- Steinberg F.M., Holt R.R., Schmitz H.H., Keen C.L. Cocoa procyanidin chain length does not determine ability to protect LDL from oxidation when monomer units are controlled. J. Nutr. Biochem. 2002;13:645–652. - PubMed
-
- Arts I.C.W., Hollman P.C.H., Feskens E.J. M., Bueno de Mesquita H.B., Kromhout D. Catechin intake might explain the inverse relation between tea consumption and ischemic heart disease: the Zutphen Elderly Study. Am. J. Clin. Nutr. 2001;74:227–232. - PubMed
-
- Arts I.C.W., Jacobs D.R., Jr, Harnack L.J., Gross M., Folsom A.R. Dietary catechins in relation to coronary heart disease death among postmenopausal women. Epidemiology. 2001;12:668–675. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

