Regulation of neural KCNQ channels: signalling pathways, structural motifs and functional implications
- PMID: 18238808
- PMCID: PMC2375728
- DOI: 10.1113/jphysiol.2007.148304
Regulation of neural KCNQ channels: signalling pathways, structural motifs and functional implications
Abstract
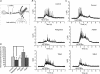
Neural M-type (KCNQ/Kv7) K(+) channels control somatic excitability, bursting and neurotransmitter release throughout the nervous system. Their activity is regulated by multiple signalling pathways. In superior cervical ganglion sympathetic neurons, muscarinic M(1), angiotensin II AT(1), bradykinin B(2) and purinergic P2Y agonists suppress M current (I(M)). Probes of PLC activity show agonists of all four receptors to induce robust PIP(2) hydrolysis. We have grouped these receptors into two related modes of action. One mode involves depletion of phosphatidylinositol 4,5-bisphosphate (PIP(2)) in the membrane, whose interaction with the channels is thought necessary for their function. The other involves IP(3)-mediated intracellular Ca(2+) signals that stimulate PIP(2) synthesis, preventing its depletion, and suppress I(M) via calmodulin. Carbon-fibre amperometry can evaluate the effect of M channel activity on release of neurotransmitter. Consistent with the dominant role of M current in control of neuronal discharge, M channel openers, or blockers, reduced or augmented the evoked release of noradrenaline neurotransmitter from superior cervical ganglion (SCG) neurons, respectively. We seek to localize the subdomains on the channels critical to their regulation by PIP(2). Based on single-channel recordings from chimeras between high-PIP(2) affinity KCNQ3 and low-PIP(2) affinity KCNQ4 channels, we focus on a 57-residue domain within the carboxy-terminus that is a possible PIP(2) binding site. Homology modelling of this domain using the published structure of IRK1 channels as a template predicts a structure very similar to an analogous region in IRK1 channels, and shows a cluster of basic residues in the KCNQ2 domain to correspond to those implicated in PIP(2) regulation of Kir channels. We discuss some important issues dealing with these topics.
Figures


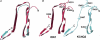
References
-
- Awatramani GB, Price GD, Trussell LO. Modulation of transmitter release by presynaptic resting potential and background calcium levels. Neuron. 2005;48:109–121. - PubMed
-
- Balla A, Balla T. Phosphatidylinositol 4-kinases: old enzymes with emerging functions. Trends Cell Biol. 2006;16:351–361. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

