Structural analysis of bikunin glycosaminoglycan
- PMID: 18247611
- PMCID: PMC4117376
- DOI: 10.1021/ja0778500
Structural analysis of bikunin glycosaminoglycan
Abstract
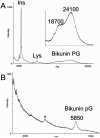
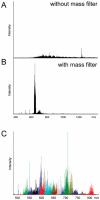
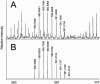
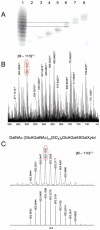
The structure of an intact glycosaminoglycan (GAG) chain of the bikunin proteoglycan (PG) was analyzed using a combined top-down and bottom-up sequencing strategy. PGs are proteins with one or more linear, high-molecular weight, sulfated GAG polysaccharides O-linked to serine or threonine residues. GAGs are often responsible for the biological functions of PGs, and subtle variations in the GAG structure have pronounced physiological effects. Bikunin is a serine protease inhibitor found in human amniotic fluid, plasma, and urine. Bikunin is posttranslationally modified with a chondroitin sulfate (CS) chain, O-linked to a serine residue of the core protein. Recent studies have shown that the CS chain of bikunin plays an important role in the physiological and pathological functions of this PG. While no PG or GAG has yet been sequenced, bikunin, the least complex PG, offers a compelling target. Electrospray ionization Fourier transform-ion cyclotron resonance mass spectrometry (ESI FTICR-MS) permitted the identification of several major components in the GAG mixture having molecular masses in a range of 5505-7102 Da. This is the first report of a mass spectrum of an intact GAG component of a PG. FTICR-MS analysis of a size-uniform fraction of bikunin GAG mixture obtained by preparative polyacrylamide gel electrophoresis, allowed the determination of chain length and number of sulfo groups in the intact GAGs.
Figures
Similar articles
-
The proteoglycan bikunin has a defined sequence.Nat Chem Biol. 2011 Oct 9;7(11):827-33. doi: 10.1038/nchembio.673. Nat Chem Biol. 2011. PMID: 21983600 Free PMC article.
-
Organization of the inter-alpha-inhibitor heavy chains on the chondroitin sulfate originating from Ser(10) of bikunin: posttranslational modification of IalphaI-derived bikunin.Biochemistry. 1999 Sep 7;38(36):11804-13. doi: 10.1021/bi9908540. Biochemistry. 1999. PMID: 10512637
-
Electrospray ionization Fourier transform mass spectrometric analysis of intact bikunin glycosaminoglycan from normal human plasma.Int J Mass Spectrom. 2011 Aug 15;305(2-3):109-115. doi: 10.1016/j.ijms.2010.09.020. Int J Mass Spectrom. 2011. PMID: 21860600 Free PMC article.
-
Applications of capillary electrophoresis electrospray ionization mass spectrometry in glycosaminoglycan analysis.Electrophoresis. 2016 Apr;37(7-8):973-86. doi: 10.1002/elps.201500461. Epub 2016 Feb 1. Electrophoresis. 2016. PMID: 26701317 Review.
-
Modern developments in mass spectrometry of chondroitin and dermatan sulfate glycosaminoglycans.Amino Acids. 2011 Jul;41(2):235-56. doi: 10.1007/s00726-010-0682-4. Epub 2010 Jul 15. Amino Acids. 2011. PMID: 20632047 Review.
Cited by
-
Proteoglycomics: recent progress and future challenges.OMICS. 2010 Aug;14(4):389-99. doi: 10.1089/omi.2009.0123. OMICS. 2010. PMID: 20450439 Free PMC article. Review.
-
Irreversible heavy chain transfer to chondroitin.J Biol Chem. 2014 Oct 17;289(42):29171-9. doi: 10.1074/jbc.M114.600809. Epub 2014 Aug 18. J Biol Chem. 2014. PMID: 25135638 Free PMC article.
-
Heparin sensing: blue-chip binding.Nat Chem. 2013 Aug;5(8):644-6. doi: 10.1038/nchem.1701. Epub 2013 Jul 7. Nat Chem. 2013. PMID: 23881491 Free PMC article.
-
Analysis of heparin oligosaccharides by capillary electrophoresis-negative-ion electrospray ionization mass spectrometry.Anal Bioanal Chem. 2017 Jan;409(2):411-420. doi: 10.1007/s00216-016-9662-1. Epub 2016 Jun 20. Anal Bioanal Chem. 2017. PMID: 27325464 Free PMC article.
-
A Retrospective Analysis of the Cartilage Kunitz Protease Inhibitory Proteins Identifies These as Members of the Inter-α-Trypsin Inhibitor Superfamily with Potential Roles in the Protection of the Articulatory Surface.Int J Mol Sci. 2019 Jan 24;20(3):497. doi: 10.3390/ijms20030497. Int J Mol Sci. 2019. PMID: 30678366 Free PMC article.
References
-
- Bulow HE, Hobert O. Annu. Rev. Cell Dev. Biol. 2006;22:375–407. - PubMed
-
- Handel TM, Johnson Z, Crown SE, Lau EK, Sweeney M, Proudfoot AE. Annu. Rev. Biochem. 2005;74:385–410. - PubMed
-
- Iozzo RV. Nat. Rev. Mol. Cell Biol. 2005;6:646–656. - PubMed
-
- Lamanna WC, Kalus I, Padva M, Baldwin RJ, Merry CLR, Dierks T. J Biotechnol. 2007;129:290–307. - PubMed
-
- Lin X. Development. 2004;131:6009–6021. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

