The Delta fbpA mutant derived from Mycobacterium tuberculosis H37Rv has an enhanced susceptibility to intracellular antimicrobial oxidative mechanisms, undergoes limited phagosome maturation and activates macrophages and dendritic cells
- PMID: 18248626
- PMCID: PMC3668688
- DOI: 10.1111/j.1462-5822.2008.01126.x
The Delta fbpA mutant derived from Mycobacterium tuberculosis H37Rv has an enhanced susceptibility to intracellular antimicrobial oxidative mechanisms, undergoes limited phagosome maturation and activates macrophages and dendritic cells
Abstract
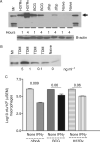
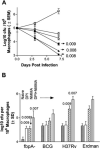
Mycobacterium tuberculosis H37Rv (Mtb) excludes phagocyte oxidase (phox) and inducible nitric oxide synthase (iNOS) while preventing lysosomal fusion in macrophages (MPhis). The antigen 85A deficient (Delta fbpA) mutant of Mtb was vaccinogenic in mice and the mechanisms of attenuation were compared with MPhis infected with H37Rv and BCG. Delta fbpA contained reduced amounts of trehalose 6, 6, dimycolate and induced minimal levels of SOCS-1 in MPhis. Blockade of oxidants enhanced the growth of Delta fbpA in MPhis that correlated with increased colocalization with phox and iNOS. Green fluorescent protein-expressing strains within MPhis or purified phagosomes were analysed for endosomal traffick with immunofluorescence and Western blot. Delta fbpA phagosomes were enriched for rab5, rab11, LAMP-1 and Hck suggesting enhanced fusion with early, recycling and late endosomes in MPhis compared with BCG or H37Rv. Delta fbpA phagosomes were thus more mature than H37Rv or BCG although, they failed to acquire rab7 and CD63 preventing lysosomal fusion. Finally, Delta fbpA infected MPhis and dendritic cells (DCs) showed an enhanced MHC-II and CD1d expression and primed immune T cells to release more IFN-gamma compared with those infected with BCG and H37Rv. Delta fbpA was thus more immunogenic in MPhis and DCs because of an enhanced susceptibility to oxidants and increased maturation.
Figures
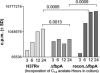

Similar articles
-
A mutant of Mycobacterium tuberculosis H37Rv that lacks expression of antigen 85A is attenuated in mice but retains vaccinogenic potential.Infect Immun. 2004 Dec;72(12):7084-95. doi: 10.1128/IAI.72.12.7084-7095.2004. Infect Immun. 2004. PMID: 15557632 Free PMC article.
-
The fbpA/sapM double knock out strain of Mycobacterium tuberculosis is highly attenuated and immunogenic in macrophages.PLoS One. 2012;7(5):e36198. doi: 10.1371/journal.pone.0036198. Epub 2012 May 4. PLoS One. 2012. PMID: 22574140 Free PMC article.
-
Processing of Mycobacterium tuberculosis antigen 85B involves intraphagosomal formation of peptide-major histocompatibility complex II complexes and is inhibited by live bacilli that decrease phagosome maturation.J Exp Med. 2001 Nov 19;194(10):1421-32. doi: 10.1084/jem.194.10.1421. J Exp Med. 2001. PMID: 11714749 Free PMC article.
-
Lipoarabinomannan, and its related glycolipids, induce divergent and opposing immune responses to Mycobacterium tuberculosis depending on structural diversity and experimental variations.Tuberculosis (Edinb). 2016 Jan;96:120-30. doi: 10.1016/j.tube.2015.09.005. Epub 2015 Oct 28. Tuberculosis (Edinb). 2016. PMID: 26586646 Review.
-
Mycobacterium tuberculosis phagosome.Mol Microbiol. 1999 Mar;31(6):1603-9. doi: 10.1046/j.1365-2958.1999.01279.x. Mol Microbiol. 1999. PMID: 10209735 Review.
Cited by
-
The cell envelope glycoconjugates of Mycobacterium tuberculosis.Crit Rev Biochem Mol Biol. 2014 Sep-Oct;49(5):361-99. doi: 10.3109/10409238.2014.925420. Epub 2014 Jun 10. Crit Rev Biochem Mol Biol. 2014. PMID: 24915502 Free PMC article. Review.
-
Lactoferrin Augmentation of the BCG Vaccine Leads to Increased Pulmonary Integrity.Tuberc Res Treat. 2011;2011:835410. doi: 10.1155/2011/835410. Epub 2011 Apr 28. Tuberc Res Treat. 2011. PMID: 22567270 Free PMC article.
-
Analysis of phagosomal proteomes: from latex-bead to bacterial phagosomes.Proteomics. 2010 Nov;10(22):4098-116. doi: 10.1002/pmic.201000210. Proteomics. 2010. PMID: 21080496 Free PMC article. Review.
-
Characterisation of iunH gene knockout strain from Mycobacterium tuberculosis.Mem Inst Oswaldo Cruz. 2017 Mar;112(3):203-208. doi: 10.1590/0074-02760160462. Mem Inst Oswaldo Cruz. 2017. PMID: 28225907 Free PMC article.
-
Pathogenic mycobacteria achieve cellular persistence by inhibiting the Niemann-Pick Type C disease cellular pathway.Wellcome Open Res. 2016 Nov 18;1:18. doi: 10.12688/wellcomeopenres.10036.2. Wellcome Open Res. 2016. PMID: 28008422 Free PMC article.
References
-
- Astarie-Dequeker C, Carreno S, Cougoule C, Maridonneau-Parini I. The protein tyrosine kinase Hck is located on lysosomal vesicles that are physically and functionally distinct from CD63-positive lysosomes in human macrophages. J Cell Sci. 2002;115:81–89. - PubMed
-
- Babior BM. NADPH oxidase: an update. Blood. 1999;93:1464–1476. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

