The Timothy syndrome mutation differentially affects voltage- and calcium-dependent inactivation of CaV1.2 L-type calcium channels
- PMID: 18250309
- PMCID: PMC2538892
- DOI: 10.1073/pnas.0710501105
The Timothy syndrome mutation differentially affects voltage- and calcium-dependent inactivation of CaV1.2 L-type calcium channels
Abstract
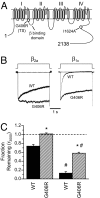
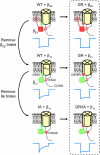
Calcium entry into excitable cells is an important physiological signal, supported by and highly sensitive to the activity of voltage-gated Ca2+ channels. After membrane depolarization, Ca2+ channels first open but then undergo various forms of negative feedback regulation including voltage- and calcium-dependent inactivation (VDI and CDI, respectively). Inactivation of Ca2+ channel activity is perturbed in a rare yet devastating disorder known as Timothy syndrome (TS), whose features include autism or autism spectrum disorder along with severe cardiac arrhythmia and developmental abnormalities. Most cases of TS arise from a sporadic single nucleotide change that generates a mutation (G406R) in the pore-forming subunit of the L-type Ca2+ channel Ca(V)1.2. We found that the TS mutation powerfully and selectively slows VDI while sparing or possibly speeding the kinetics of CDI. The deceleration of VDI was observed when the L-type channels were expressed with beta1 subunits prominent in brain, as well as beta2 subunits of importance for the heart. Dissociation of VDI and CDI was further substantiated by measurements of Ca2+ channel gating currents and by analysis of another channel mutation (I1624A) that hastens VDI, acting upstream of the step involving Gly406. As highlighted by the TS mutation, CDI does not proceed to completeness but levels off at approximately 50%, consistent with a change in gating modes and not an absorbing inactivation process. Thus, the TS mutation offers a unique perspective on mechanisms of inactivation as well as a promising starting point for exploring the underlying pathophysiology of autism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Reichenbach H, Meister EM, Theile H. Kinderarztl Prax. 1992;60:54–56. - PubMed
-
- Marks ML, Trippel DL, Keating MT. Am J Cardiol. 1995;76:744–745. - PubMed
-
- Marks ML, Whisler SL, Clericuzio C, Keating M. J Am Coll Cardiol. 1995;25:59–64. - PubMed
-
- Splawski I, Timothy KW, Sharpe LM, Decher N, Kumar P, Bloise R, Napolitano C, Schwartz PJ, Joseph RM, Condouris K, et al. Cell. 2004;119:19–31. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

