Mitochondrial lactate dehydrogenase is involved in oxidative-energy metabolism in human astrocytoma cells (CCF-STTG1)
- PMID: 18253497
- PMCID: PMC2212712
- DOI: 10.1371/journal.pone.0001550
Mitochondrial lactate dehydrogenase is involved in oxidative-energy metabolism in human astrocytoma cells (CCF-STTG1)
Abstract
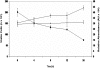
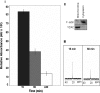
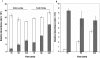
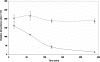
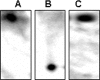
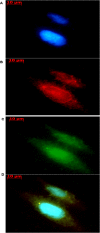
Lactate has long been regarded as an end product of anaerobic energy production and its fate in cerebral metabolism has not been precisely delineated. In this report, we demonstrate, for the first time, the ability of a human astrocytic cell line (CCF-STTG1) to consume lactate and to generate ATP via oxidative phosphorylation. (13)C-NMR and HPLC analyses aided in the identification of tricarboxylic acid (TCA) cyle metabolites and ATP in the astrocytic mitochondria incubated with lactate. Oxamate, an inhibitor of lactate dehydrogenase (LDH), abolished mitochondrial lactate consumption. Electrophoretic and fluorescence microscopic analyses helped localize LDH in the mitochondria. Taken together, this study implicates lactate as an important contributor to ATP metabolism in the brain, a finding that may significantly change our notion of how this important organ manipulates its energy budget.
Conflict of interest statement
Figures




Similar articles
-
The cellular and compartmental profile of mouse retinal glycolysis, tricarboxylic acid cycle, oxidative phosphorylation, and ~P transferring kinases.Mol Vis. 2016 Jul 23;22:847-85. eCollection 2016. Mol Vis. 2016. PMID: 27499608 Free PMC article.
-
Mitochondrial lactate metabolism is involved in antioxidative defense in human astrocytoma cells.J Neurosci Res. 2014 Apr;92(4):464-75. doi: 10.1002/jnr.23338. Epub 2014 Jan 22. J Neurosci Res. 2014. PMID: 24452607
-
Mitochondrial fission induces glycolytic reprogramming in cancer-associated myofibroblasts, driving stromal lactate production, and early tumor growth.Oncotarget. 2012 Aug;3(8):798-810. doi: 10.18632/oncotarget.574. Oncotarget. 2012. PMID: 22878233 Free PMC article.
-
Including the mitochondrial metabolism of L-lactate in cancer metabolic reprogramming.Cell Mol Life Sci. 2018 Aug;75(15):2763-2776. doi: 10.1007/s00018-018-2831-y. Epub 2018 May 4. Cell Mol Life Sci. 2018. PMID: 29728715 Free PMC article. Review.
-
Enzymes involved in l-lactate metabolism in humans.Mitochondrion. 2013 Nov;13(6):615-29. doi: 10.1016/j.mito.2013.08.011. Epub 2013 Sep 9. Mitochondrion. 2013. PMID: 24029012 Review.
Cited by
-
ATM-NFκB axis-driven TIGAR regulates sensitivity of glioma cells to radiomimetics in the presence of TNFα.Cell Death Dis. 2013 May 2;4(5):e615. doi: 10.1038/cddis.2013.128. Cell Death Dis. 2013. PMID: 23640457 Free PMC article.
-
Intracellular shuttle: the lactate aerobic metabolism.ScientificWorldJournal. 2012;2012:420984. doi: 10.1100/2012/420984. Epub 2012 Apr 19. ScientificWorldJournal. 2012. PMID: 22593684 Free PMC article.
-
Involvement of the two L-lactate dehydrogenase in development and pathogenicity in Fusarium graminearum.Curr Genet. 2019 Apr;65(2):591-605. doi: 10.1007/s00294-018-0909-6. Epub 2018 Nov 24. Curr Genet. 2019. PMID: 30474697
-
Mitochondria-associated lactate dehydrogenase is not a biologically significant contributor to bioenergetic function in murine striated muscle.Redox Biol. 2019 Jun;24:101177. doi: 10.1016/j.redox.2019.101177. Epub 2019 Mar 22. Redox Biol. 2019. PMID: 30939431 Free PMC article.
-
Glutamate levels and perfusion in pons during migraine attacks: A 3T MRI study using proton spectroscopy and arterial spin labeling.J Cereb Blood Flow Metab. 2021 Mar;41(3):604-616. doi: 10.1177/0271678X20906902. Epub 2020 May 19. J Cereb Blood Flow Metab. 2021. PMID: 32423331 Free PMC article. Clinical Trial.
References
-
- Bergersen LH. Is lactate food for neurons? Comparison of monocarboxylate transporter subtypes in brain and muscle. Neuroscience. 2007;145:11–19. - PubMed
-
- Brooks GA. Lactate production under fully aerobic conditions: the lactate shuttle during rest and exercise. Fed Proc. 1986;45:2924–2929. - PubMed
-
- Brooks GA. Mammalian fuel utilization during sustained exercise. Comp Biochem Physiol B Biochem Mol Biol. 1998;120:89–107. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

