Recovery of humoral immunity is critical for successful antiviral therapy in disseminated mouse adenovirus type 1 infection
- PMID: 18268085
- PMCID: PMC2292527
- DOI: 10.1128/AAC.01311-07
Recovery of humoral immunity is critical for successful antiviral therapy in disseminated mouse adenovirus type 1 infection
Abstract
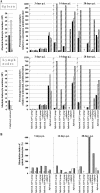
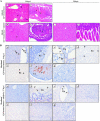
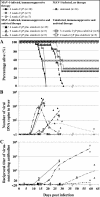
Severe adenovirus infections in transplant recipients undergoing immunosuppressive therapy are of increasing concern. Controversy exists on the contribution of antiviral therapy and the host immune response to recovery from these infections. Here, we established a systemic mouse adenovirus type 1 (MAV-1) infection in cyclophosphamide (CyP)-treated BALB/c mice. CyP was administered at 100 mg per kg of body weight every other day for 2, 3, or 4 weeks, thereby inducing general but reversible leukopenia, with a major suppression of the B-cell numbers and functionality that was more pronounced than that seen with T cells. The outcome of MAV-1 infection was dependent on the duration of CyP therapy, as the mice with the most severe immunosuppression were the most vulnerable to MAV-1-induced hemorrhagic enteritis and mortality. The protective effect of concomitant antiviral therapy with cidofovir depended on the level of immunosuppression. The combination of cidofovir treatment with the withdrawal of immunosuppression was the most successful regimen for increasing survival rates. Survival was clearly correlated with the clearance of virus and increased titers of MAV-1-specific antibodies in sera. In addition, the passive transfer of MAV-1-specific immunoglobulin G into MAV-1-infected SCID BALB/c mice caused a marked delay in mortality, the extent of the delay being dependent on the titer of MAV-1-specific antibodies. Based on the critical role of the humoral immune response in the early defense against disseminated adenovirus infection, the concomitant use of adenovirus-specific immunoglobulins and antiviral therapy should be considered for transplant patients at risk for severe adenovirus infections.
Figures


Similar articles
-
Mouse adenovirus type 1 infection in SCID mice: an experimental model for antiviral therapy of systemic adenovirus infections.Antimicrob Agents Chemother. 2005 Nov;49(11):4689-99. doi: 10.1128/AAC.49.11.4689-4699.2005. Antimicrob Agents Chemother. 2005. PMID: 16251313 Free PMC article.
-
Safety and efficacy of CMX001 as salvage therapy for severe adenovirus infections in immunocompromised patients.Biol Blood Marrow Transplant. 2012 May;18(5):731-8. doi: 10.1016/j.bbmt.2011.09.007. Epub 2011 Sep 29. Biol Blood Marrow Transplant. 2012. PMID: 21963623 Free PMC article.
-
Evaluation of Cidofovir (HPMPC, GS-504) against adenovirus type 5 infection in vitro and in a New Zealand rabbit ocular model.Antiviral Res. 1996 Jul;31(3):165-72. doi: 10.1016/0166-3542(95)00962-0. Antiviral Res. 1996. PMID: 8811201
-
Disseminated adenovirus infection after allogeneic stem cell transplant and the potential role of brincidofovir - Case series and 10 year experience of management in an adult transplant cohort.J Clin Virol. 2017 Nov;96:73-79. doi: 10.1016/j.jcv.2017.09.013. Epub 2017 Oct 3. J Clin Virol. 2017. PMID: 29017084 Review.
-
Intravenous ribavirin treatment for severe adenovirus disease in immunocompromised children.Pediatrics. 2002 Jul;110(1 Pt 1):e9. doi: 10.1542/peds.110.1.e9. Pediatrics. 2002. PMID: 12093990 Review.
Cited by
-
Small-size recombinant adenoviral hexon protein fragments for the production of virus-type specific antibodies.Virol J. 2017 Aug 18;14(1):158. doi: 10.1186/s12985-017-0822-5. Virol J. 2017. PMID: 28821267 Free PMC article.
-
Murine adenoviruses: tools for studying adenovirus pathogenesis in a natural host.FEBS Lett. 2019 Dec;593(24):3649-3659. doi: 10.1002/1873-3468.13699. Epub 2019 Dec 6. FEBS Lett. 2019. PMID: 31777948 Free PMC article. Review.
-
The RGD-binding integrins αvβ6 and αvβ8 are receptors for mouse adenovirus-1 and -3 infection.PLoS Pathog. 2021 Dec 15;17(12):e1010083. doi: 10.1371/journal.ppat.1010083. eCollection 2021 Dec. PLoS Pathog. 2021. PMID: 34910784 Free PMC article.
-
Ganciclovir inhibits human adenovirus replication and pathogenicity in permissive immunosuppressed Syrian hamsters.Antimicrob Agents Chemother. 2014 Dec;58(12):7171-81. doi: 10.1128/AAC.03860-14. Epub 2014 Sep 15. Antimicrob Agents Chemother. 2014. PMID: 25224011 Free PMC article.
-
Large-Scale Analysis of Drug Side Effects via Complex Regulatory Modules Composed of microRNAs, Transcription Factors and Gene Sets.Sci Rep. 2017 Jul 20;7(1):5962. doi: 10.1038/s41598-017-06083-5. Sci Rep. 2017. PMID: 28729650 Free PMC article.
References
-
- Alinari, L., R. Lapalombella, L. Andritsos, R. A. Baiocchi, T. S. Lin, and J. C. Byrd. 2007. Alemtuzumab (Campath-1H) in the treatment of chronic lymphocytic leukemia. Oncogene 26:3644-3653. - PubMed
-
- Angulo, I., F. G. las Heras, J. F. Garcia-Bustos, D. Gargallo, M. A. Munoz-Fernandez, and M. Fresno. 2000. Nitric oxide-producing CD11b+ Ly-6G(Gr-1)+ CD31(ER-MP12)+ cells in the spleen of cyclophosphamide-treated mice: implications for T-cell responses in immunosuppressed mice. Blood 95:212-220. - PubMed
-
- Avivi, I., S. Chakrabarti, D. W. Milligan, H. Waldmann, G. Hale, H. Osman, K. N. Ward, C. D. Fegan, K. Yong, A. H. Goldstone, D. C. Linch, and S. Mackinnon. 2004. Incidence and outcome of adenovirus disease in transplant recipients after reduced-intensity conditioning with alemtuzumab. Biol. Blood Marrow Transplant. 10:186-194. - PubMed
-
- Bordigoni, P., A. S. Carret, V. Venard, F. Witz, and A. Le Faou. 2001. Treatment of adenovirus infections in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin. Infect. Dis. 32:1290-1297. - PubMed
-
- Cao, L., A. Martin, N. Polakos, and J. A. Moynihan. 2004. Stress causes a further decrease in immunity to herpes simplex virus-1 in immunocompromised hosts. J. Neuroimmunol. 156:21-30. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

