Helical domain and kinase domain mutations in p110alpha of phosphatidylinositol 3-kinase induce gain of function by different mechanisms
- PMID: 18268322
- PMCID: PMC2268191
- DOI: 10.1073/pnas.0712169105
Helical domain and kinase domain mutations in p110alpha of phosphatidylinositol 3-kinase induce gain of function by different mechanisms
Abstract
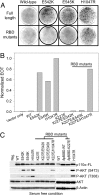
The phosphatidylinositol 3-kinase (PI3K) signaling pathway is up-regulated in cancer. PIK3CA, the gene coding for the catalytic subunit p110alpha of PI3K, is mutated in approximately 30% of tumors of the prostate, breast, cervix, and endometrium. The most prominent of these mutants, represented by single amino acid substitutions in the helical or kinase domain, show a gain of enzymatic function, activate AKT signaling, and induce oncogenic transformation. We have carried out a genetic and biochemical analysis of these hot-spot mutations in PIK3CA. The results of this study suggest that the helical and kinase domain mutations trigger gain of function through different mechanisms. They show different requirements for interaction with the PI3K regulatory subunit p85 and with RAS-GTP. The gain of function induced by helical domain mutations is independent of binding to p85 but requires interaction with RAS-GTP. In contrast, the kinase domain mutation is active in the absence of RAS-GTP binding but is highly dependent on the interaction with p85. We speculate that the contrasting roles of p85 and RAS-GTP in helical and kinase domain mutations reflect two distinct states of mutated p110alpha. These two states differ in mutation-induced surface charges and also may differ in conformational properties that are controlled by interactions with p85 and RAS-GTP. The two states do not appear mutually exclusive because the helical and kinase domain mutations act synergistically when present in the same p110alpha molecule. This synergism also supports the conclusion that the helical and kinase domain mutations operate by two different and independent mechanisms.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
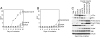

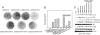

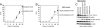
References
-
- Bader AG, Kang S, Zhao L, Vogt PK. Oncogenic PI3K deregulates transcription and translation. Nat Rev Cancer. 2005;5:921–929. - PubMed
-
- Bachman KE, et al. The PIK3CA gene is mutated with high frequency in human breast cancers. Cancer Biol Ther. 2004;3:772–775. - PubMed
-
- Broderick DK, et al. Mutations of PIK3CA in anaplastic oligodendrogliomas, high-grade astrocytomas, and medulloblastomas. Cancer Res. 2004;64:5048–5050. - PubMed
-
- Campbell IG, et al. Mutation of the PIK3CA gene in ovarian and breast cancer. Cancer Res. 2004;64:7678–7681. - PubMed
-
- Hartmann C, et al. PIK3CA mutations in glioblastoma multiforme. Acta Neuropathol. 2005;109:639–642. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

