Red blood cells induce hypoxic lung inflammation
- PMID: 18270324
- PMCID: PMC2384143
- DOI: 10.1182/blood-2007-09-113902
Red blood cells induce hypoxic lung inflammation
Abstract
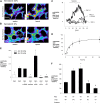
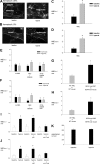
Hypoxia, which commonly associates with respiratory and cardiovascular diseases, provokes an acute inflammatory response. However, underlying mechanisms are not well understood. Here we report that red blood cells (RBCs) induce hypoxic inflammation by producing reactive oxygen species (ROS) that diffuse to endothelial cells of adjoining blood vessels. Real-time fluorescence imaging of rat and mouse lungs revealed that in the presence of RBC-containing vascular perfusion, hypoxia increased microvascular ROS, and cytosolic Ca(2+), leading to P-selectin-dependent leukocyte recruitment. However, in the presence of RBC-free perfusion, all hypoxia-induced responses were completely inhibited. Because hemoglobin (Hb) autoxidation causes RBC superoxide formation that readily dismutates to H(2)O(2), hypoxia-induced responses were lost when we inhibited Hb autoxidation with CO or nitrite, or when the H(2)O(2) inhibitor, catalase was added to the infusion to neutralize the RBC-derived ROS. By contrast, perfusion with RBCs from BERK-trait mice that are more susceptible to Hb autoxidation and to hypoxia-induced superoxide production enhanced the hypoxia-induced responses. We conclude that in hypoxia, increased Hb autoxidation augments superoxide production in RBCs. Consequently, RBCs release H(2)O(2) that diffuses to the lung microvascular endothelium, thereby initiating Ca(2+)-dependent leukocyte recruitment. These findings are the first evidence that RBCs contribute to hypoxia-induced inflammation.
Figures



Comment in
-
Hypoxic erythrocytes spark lung leukocyte adhesion.Blood. 2008 May 15;111(10):4831-2. doi: 10.1182/blood-2008-02-140947. Blood. 2008. PMID: 18467598 Free PMC article.
References
-
- Madjdpour C, Jewell UR, Kneller S, et al. Decreased alveolar oxygen induces lung inflammation. Am J Physiol Lung Cell Mol Physiol. 2003;284:L360–L367. - PubMed
-
- Peyssonnaux C, Cejudo-Martin P, Doedens A, Zinkernagel AS, Johnson RS, Nizet V. Cutting edge: essential role of hypoxia inducible factor-1alpha in development of lipopolysaccharide-induced sepsis. J Immunol. 2007;178:7516–7519. - PubMed
-
- Ichimura H, Parthasarathi K, Issekutz AC, Bhattacharya J. Pressure-induced leukocyte margination in lung postcapillary venules. Am J Physiol Lung Cell Mol Physiol. 2005;289:L407–L412. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

