Ternary Kv4.2 channels recapitulate voltage-dependent inactivation kinetics of A-type K+ channels in cerebellar granule neurons
- PMID: 18276729
- PMCID: PMC2465190
- DOI: 10.1113/jphysiol.2007.150540
Ternary Kv4.2 channels recapitulate voltage-dependent inactivation kinetics of A-type K+ channels in cerebellar granule neurons
Abstract
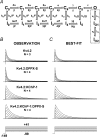
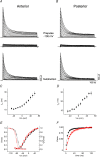
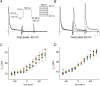
Kv4 channels mediate most of the somatodendritic subthreshold operating A-type current (I(SA)) in neurons. This current plays essential roles in the regulation of spike timing, repetitive firing, dendritic integration and plasticity. Neuronal Kv4 channels are thought to be ternary complexes of Kv4 pore-forming subunits and two types of accessory proteins, Kv channel interacting proteins (KChIPs) and the dipeptidyl-peptidase-like proteins (DPPLs) DPPX (DPP6) and DPP10. In heterologous cells, ternary Kv4 channels exhibit inactivation that slows down with increasing depolarization. Here, we compared the voltage dependence of the inactivation rate of channels expressed in heterologous mammalian cells by Kv4.2 proteins with that of channels containing Kv4.2 and KChIP1, Kv4.2 and DPPX-S, or Kv4.2, KChIP1 and DPPX-S, and found that the relation between inactivation rate and membrane potential is distinct for these four conditions. Moreover, recordings from native neurons showed that the inactivation kinetics of the I(SA) in cerebellar granule neurons has voltage dependence that is remarkably similar to that of ternary Kv4 channels containing KChIP1 and DPPX-S proteins in heterologous cells. The fact that this complex and unique behaviour (among A-type K(+) currents) is observed in both the native current and the current expressed in heterologous cells by the ternary complex containing Kv4, DPPX and KChIP proteins supports the hypothesis that somatically recorded native Kv4 channels in neurons include both types of accessory protein. Furthermore, quantitative global kinetic modelling showed that preferential closed-state inactivation and a weakly voltage-dependent opening step can explain the slowing of the inactivation rate with increasing depolarization. Therefore, it is likely that preferential closed-state inactivation is the physiological mechanism that regulates the activity of both ternary Kv4 channel complexes and native I(SA)-mediating channels.
Figures



References
-
- Adams JP, Anderson AE, Varga AW, Dineley KT, Cook RG, Pfaffinger PJ, Sweatt JD. The A-type potassium channel Kv4.2 is a substrate for the mitogen-activated protein kinase ERK. J Neurochem. 2000;75:2277–2287. - PubMed
-
- An WF, Bowlby MR, Betty M, Cao J, Ling HP, Mendoza G, Hinson JW, Mattsson KI, Strassle BW, Trimmer JS, Rhodes KJ. Modulation of A-type potassium channels by a family of calcium sensors. Nature. 2000;403:553–556. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

